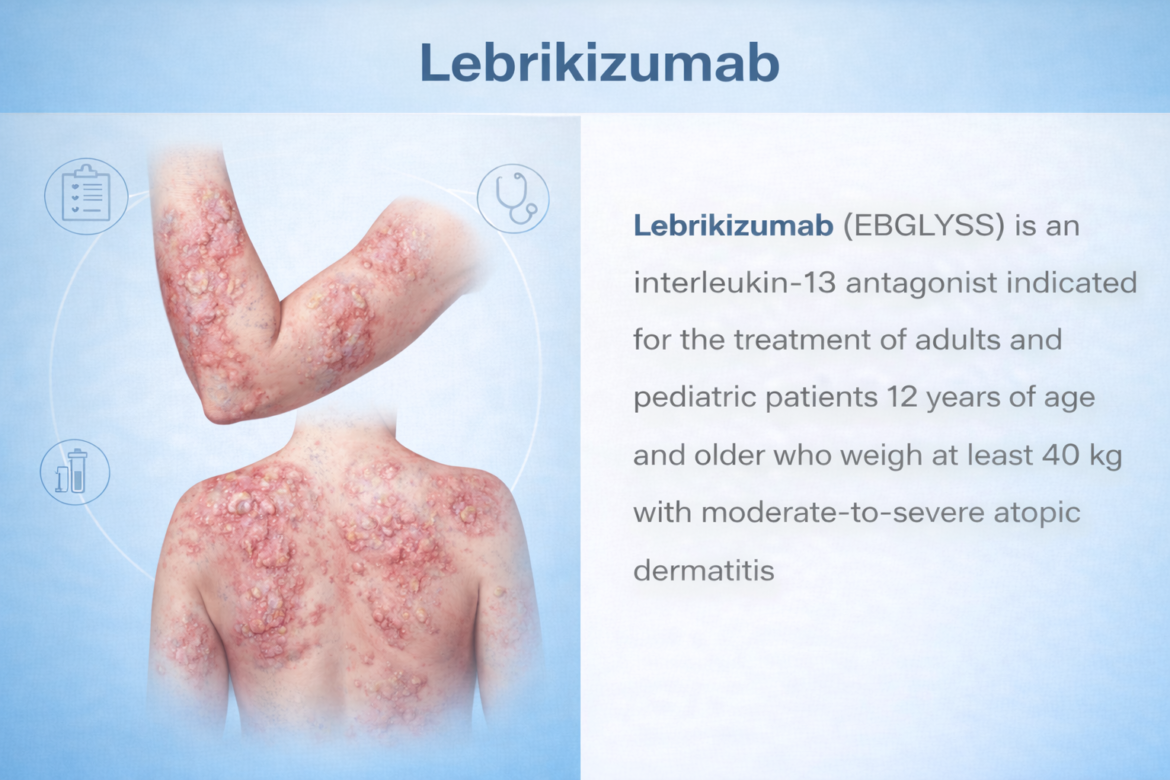
TRIALISTMD · DRUG PROFILE FDA APPROVED 2024 Anti-IL-13 Monoclonal Antibody · Atopic Dermatitis lebrikizumab EBGLYSS®
Author: trialistmd
TAPINAROF
Tapinarof (VTAMA) — TrialistMD Drug Profile TRIALISTMD · Drug Intelligence Platform FDA APPROVED 2022 AhR
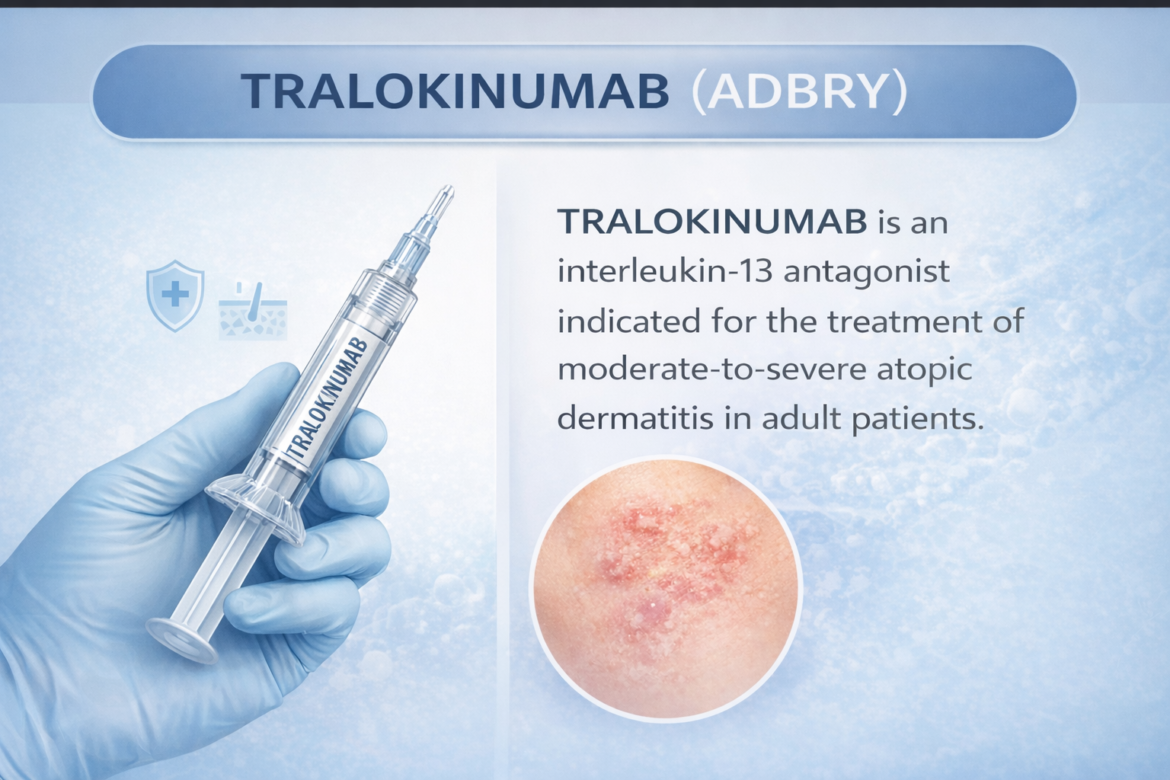
TRALOKINUMAB
Tralokinumab (ADBRY) — TrialistMD Drug Profile TRIALISTMD · Drug Intelligence Platform FDA APPROVED 2021 IL-13
DEUCRAVACITINIB
Deucravacitinib (SOTYKTU) — TrialistMD Drug Profile TRIALISTMD · Drug Intelligence Platform FDA APPROVED 2022 TYK2
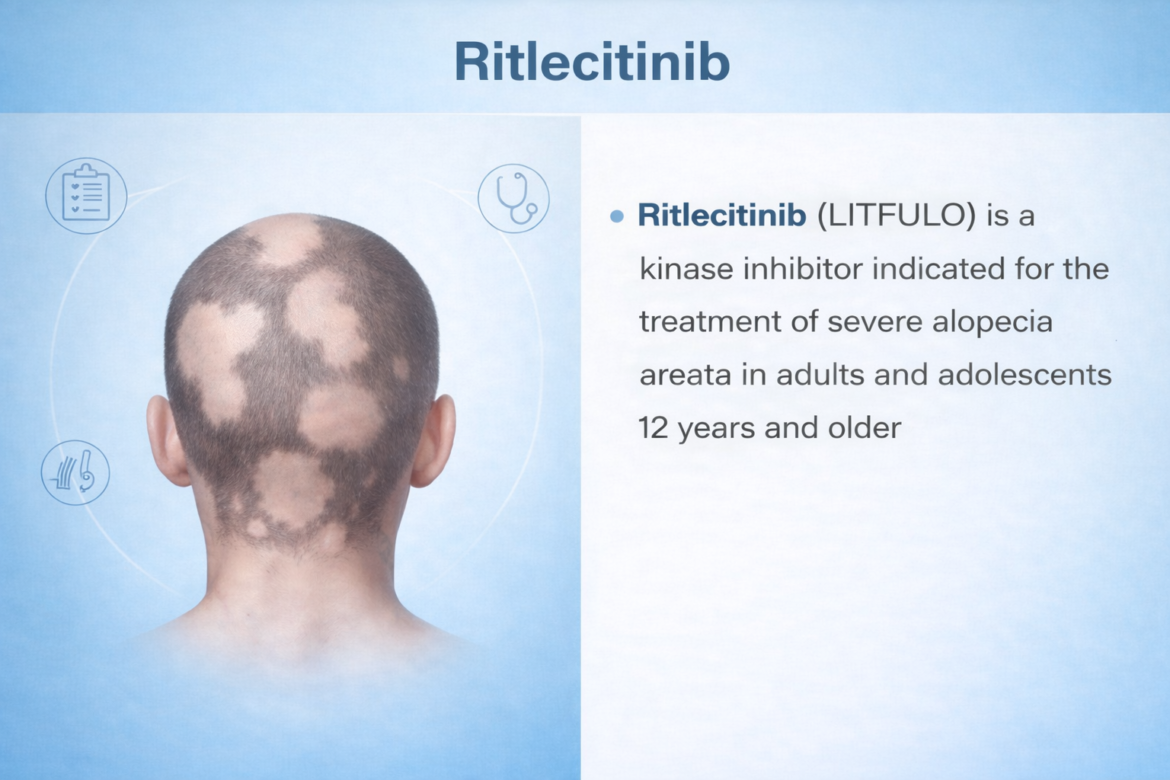
Ritlecitinib
Ritlecitinib (LITFULO) — TrialistMD Drug Profile TRIALISTMD · Drug Intelligence Platform FDA APPROVED 2023 JAK3
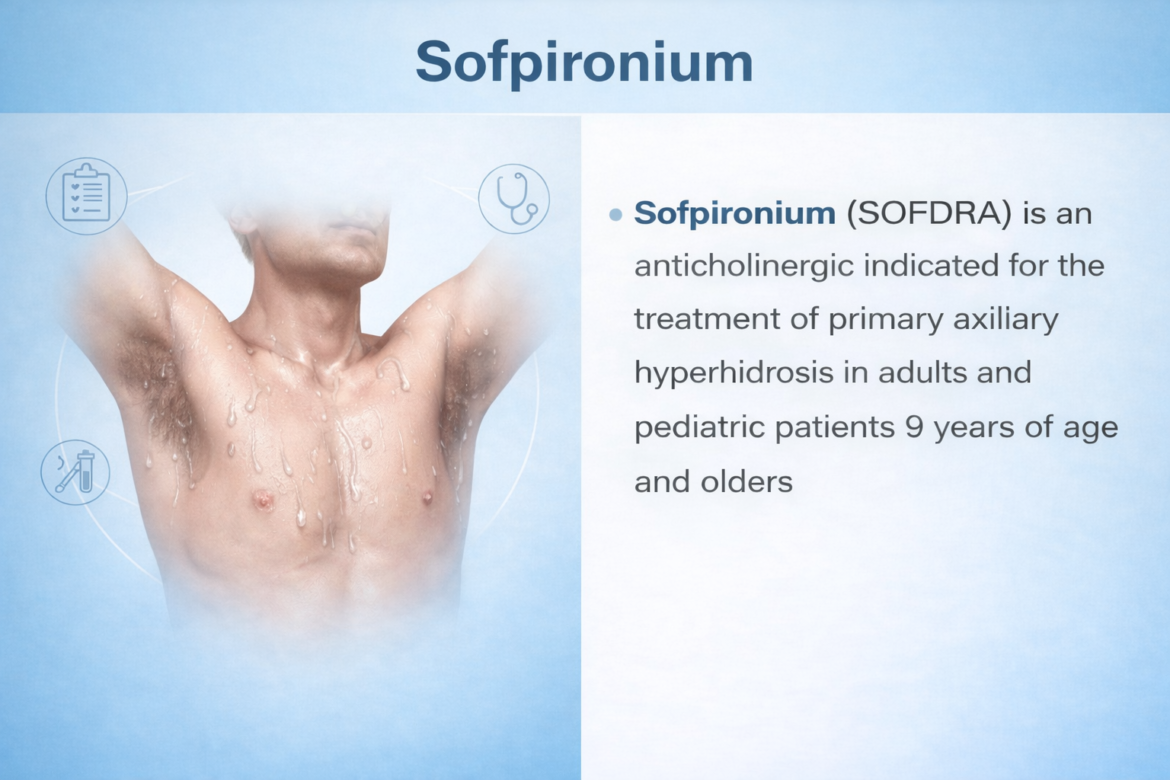
SOFPIRONIUM
Sofpironium Bromide (Sofdra) — TrialistMD Drug Profile TRIALISTMD · Drug Intelligence Platform NDA 217347 —
LETIBOTULINUMTOXIN A
LetibotulinumtoxinA-wlbg (LETYBO) – Drug Profile | TrialistMD LETYBO® letibotulinumtoxinA-wlbg FDA Approved NDA 761237 ⚠ Boxed
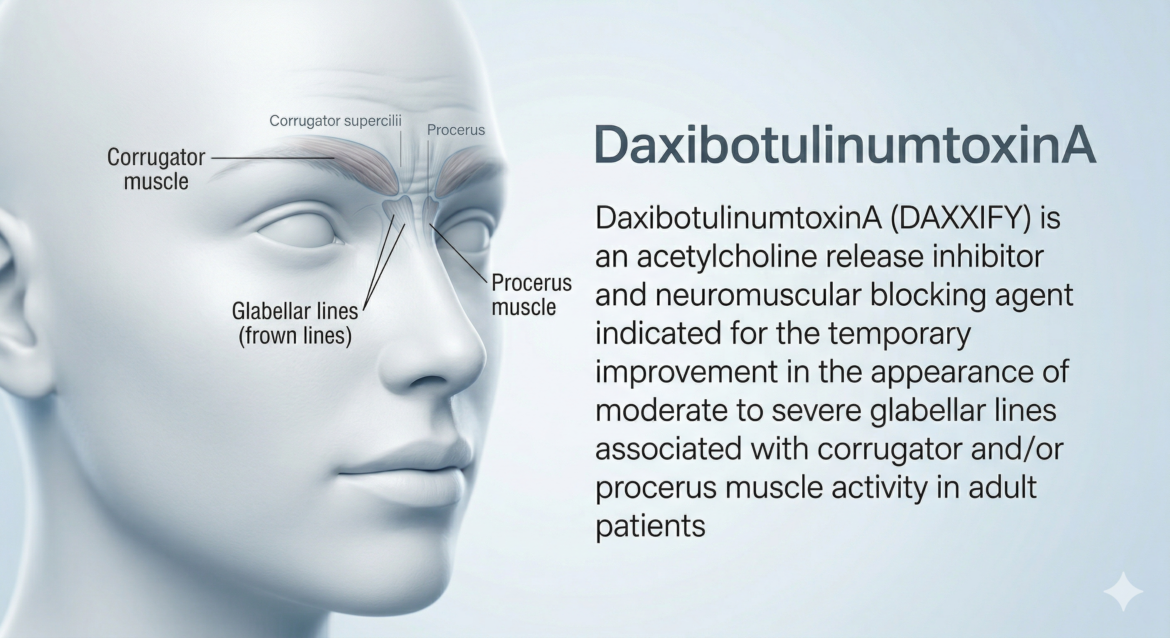
DaxibotuliumtoxinA
DaxibotulinumtoxinA-lanm (DAXXIFY) — TrialistMD Drug Profile TRIALISTMD · Drug Intelligence Platform FDA APPROVED 2022 Botulinum
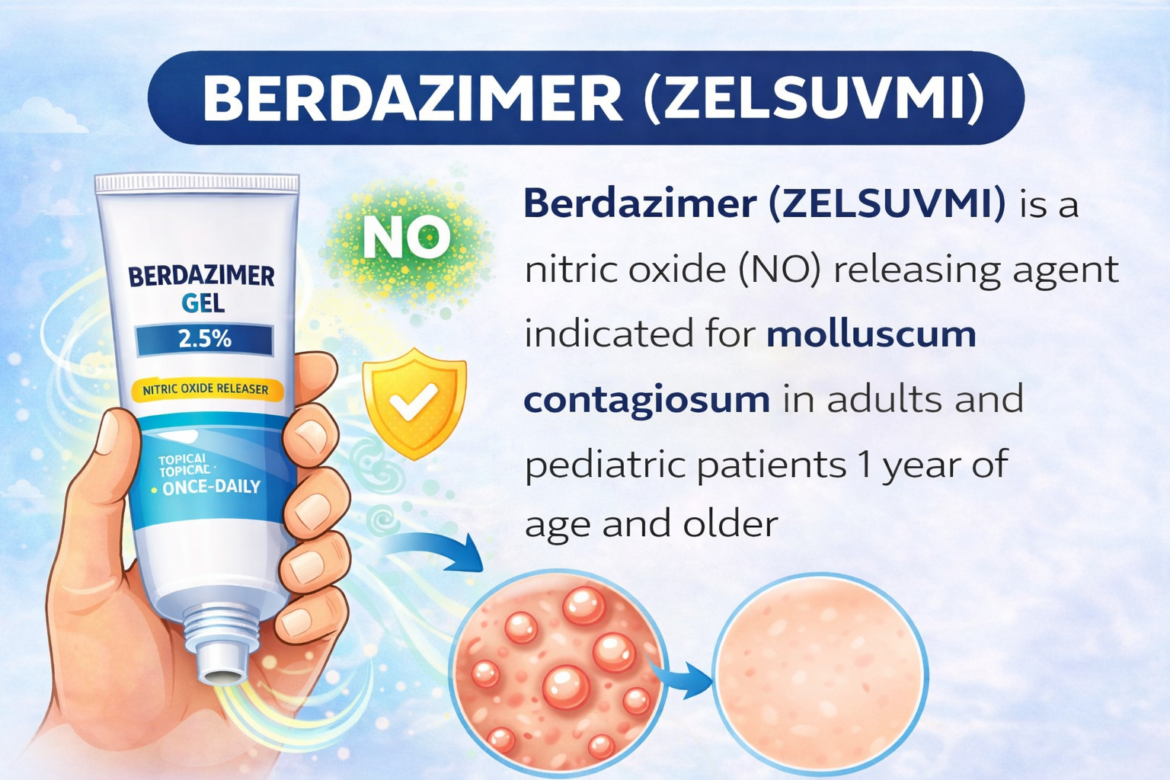
Berdazimer
Berdazimer (ZELSUVMI) — TrialistMD Drug Profile TRIALISTMD · Drug Intelligence Platform FDA APPROVED 2024 Nitric
Abrocitinib
Abrocitinib (CIBINQO) — TrialistMD Drug Profile TRIALISTMD · Drug Intelligence Platform FDA APPROVED 2022 JAK1