DaxibotulinumtoxinA-lanm
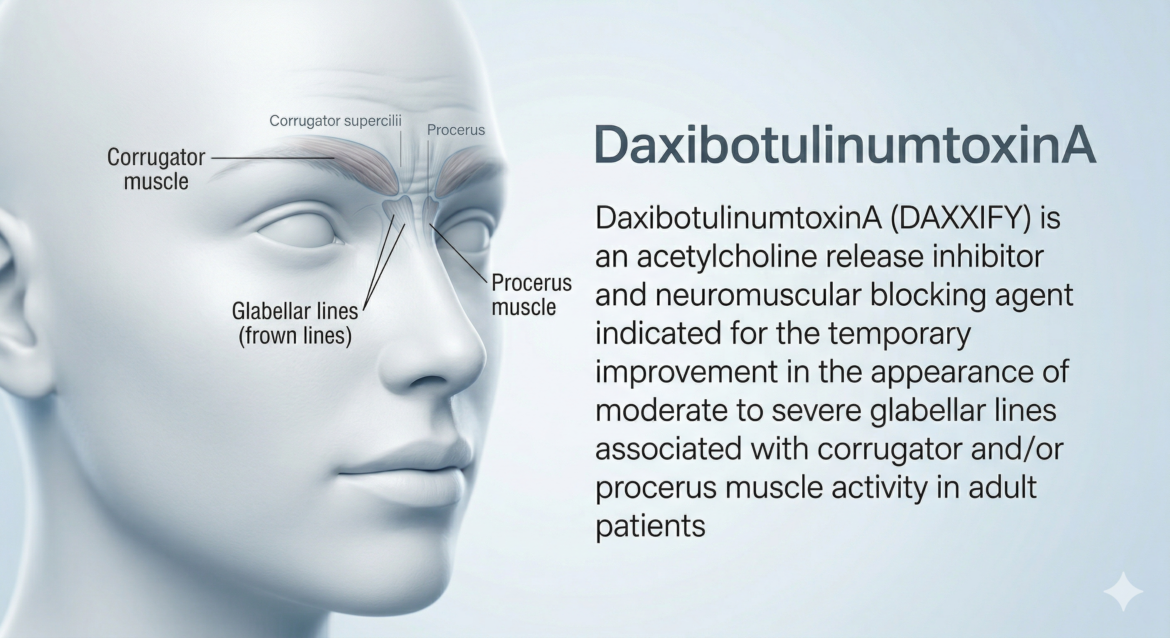
A first-in-class 150 kDa botulinum toxin type A without accessory proteins, formulated with a proprietary 35-amino acid stabilizing peptide excipient (RTP004) that confers exceptional thermal stability and extended duration of effect. The first neuromodulator to demonstrate median duration of response exceeding 6 months in clinical trials — differentiated from onabotulinumtoxinA and other BoNT-A products by its novel formulation without albumin or human serum. Carries the class Boxed Warning for distant spread of toxin effect. Approved for glabellar lines in adults only; not approved for spasticity.
Drug Overview
BLA 761127Mechanism of Action & Differentiating Features
Section 12.1 / Section 11DaxibotulinumtoxinA-lanm is a purified 150 kDa botulinum neurotoxin type A without accessory proteins, derived from Clostridium botulinum type A. Upon intramuscular injection, the toxin is internalized into the nerve terminal and translocates into the neuronal cytosol where it cleaves SNAP25 — a SNARE protein essential for synaptic vesicle membrane docking and acetylcholine release. This produces a dose-dependent decrease in muscle function. Recovery is gradual via degradation of the neurotoxin light chain and axonal sprouting, leading to eventual muscle reinnervation. The key formulation innovation is the 35-amino acid stabilizing peptide excipient RTP004, which prevents surface adsorption and promotes thermal stability, enabling room-temperature storage and the extended clinical duration observed in trials.
Competitive Landscape — Injectable Neuromodulators for Glabellar Lines (US)
| Product | INN / Toxin Type | Excipient / Stabilizer | FDA Approval (GL) | Typical Duration |
|---|---|---|---|---|
| DAXXIFY (Revance) | daxibotulinumtoxinA-lanm (BoNT-A, no accessory proteins) | RTP004 peptide (albumin-free) | Sep 8, 2022 (BLA 761127) | ~24 weeks median (pivotal trials); up to 6 months |
| BOTOX Cosmetic (AbbVie) | onabotulinumtoxinA | Human serum albumin, NaCl | Apr 15, 2002 | ~3–4 months |
| DYSPORT (Galderma) | abobotulinumtoxinA | Human serum albumin, lactose | Apr 29, 2009 | ~3–4 months |
| XEOMIN (Merz) | incobotulinumtoxinA (naked toxin) | Human serum albumin, sucrose (no complexing proteins) | Jul 20, 2011 | ~3–4 months |
| JEUVEAU (Evolus) | prabotulinumtoxinA-xvfs | Human serum albumin, NaCl | Feb 1, 2019 | ~3–4 months |
Source: FDA label BLA 761127, Ref ID 5042005; BLA 761127 Multi-disciplinary Review, Ref ID 5041522 and 5041615. Duration estimates for competitor products based on labeling and published data; not derived from head-to-head comparisons. DAXXIFY approved September 8, 2022 following resubmission in response to Complete Response Letter (manufacturing deficiencies resolved). No new clinical data were submitted in the resubmission; efficacy and safety established from first review cycle.
Baseline & Trial Characteristics
Table 12–13, BLA 761127 Medical ReviewTrial Design Summary
Section 8.1.1 / Studies 1620301 and 1620302| Parameter | SAKURA-1 (Study 1620301) | SAKURA-2 (Study 1620302) |
|---|---|---|
| Study type | Randomized, double-blind, placebo-controlled Phase 3 | Randomized, double-blind, placebo-controlled Phase 3 (identical design) |
| Randomization ratio | 2:1 (DAXXIFY:placebo) | 2:1 (DAXXIFY:placebo) |
| Sample size | N=303 (201 DAXXIFY / 102 placebo) | N=306 (204 DAXXIFY / 102 placebo) — note: 1 placebo subject treated with DAXXIFY in error |
| Dose | 40 Units (5 × 8 U IM) or placebo | 40 Units (5 × 8 U IM) or placebo |
| Follow-up | 24–36 weeks post-treatment | 24–36 weeks post-treatment |
| Assessment visits | Weeks 1, 2, 4, 8, 12, 16, 20, 24, 28, 32, 36 | Weeks 1, 2, 4, 8, 12, 16, 20, 24, 28, 32, 36 |
| Discontinuation rule | Subjects discontinued after Week 24 if IGA-FWS AND PFWS both returned to baseline | Same as SAKURA-1 |
| Primary endpoint | 2-point composite response (IGA-FWS + PFWS ≤1 AND ≥2-grade improvement) at Week 4 | Same as SAKURA-1 |
| Key secondary endpoints | IGA-FWS ≤1 and IGA-FWS + PFWS ≤1 at Weeks 2, 4, 8, 12, 16, 20, 24 | Same as SAKURA-1 |
| Statistical test | Cochran-Mantel-Haenszel, stratified by center; M-H confidence intervals | Same as SAKURA-1 |
| Missing data | “Worst/best outcome” imputation; multiple imputation (MCMC) as sensitivity analysis | Same as SAKURA-1 |
| Completion (Week 4) | 196/201 (98%) DAXXIFY; 97/102 (95%) placebo | 203/204 (99%) DAXXIFY; 99/102 (97%) placebo |
| Full study completion | 182/201 (91%) DAXXIFY; 93/102 (91%) placebo | 191/204 (94%) DAXXIFY; 93/102 (91%) placebo |
Demographics (Pooled SAKURA-1 + SAKURA-2, DAXXIFY arms)
Table 12| Characteristic | SAKURA-1 DAXXIFY (N=201) | SAKURA-2 DAXXIFY (N=204) | SAKURA-1 Placebo (N=102) | SAKURA-2 Placebo (N=102) |
|---|---|---|---|---|
| Mean age (range) | 50.9 yrs (23–74) | 49.6 yrs (21–73) | 49.0 yrs (22–74) | 50.5 yrs (27–75) |
| Age 18–45, n (%) | 58 (28.9%) | 62 (30.4%) | 32 (31.4%) | 30 (29.4%) |
| Age 46–55, n (%) | 68 (33.8%) | 91 (44.6%) | 41 (40.2%) | 42 (41.2%) |
| Age 56–75, n (%) | 75 (37.3%) | 51 (25.0%) | 29 (28.4%) | 30 (29.4%) |
| Female, n (%) | 174 (86.6%) | 183 (89.7%) | 88 (86.3%) | 87 (85.3%) |
| White, n (%) | 173 (86.1%) | 180 (88.2%) | 81 (79.4%) | 92 (90.2%) |
| Black / African American, n (%) | 10 (5.0%) | 9 (4.4%) | 8 (7.8%) | 3 (2.9%) |
| Asian, n (%) | 7 (3.5%) | 11 (5.4%) | 2 (2.0%) | 5 (4.9%) |
| Hispanic or Latino, n (%) | 47 (23.4%) | 19 (9.3%) | 25 (24.5%) | 10 (9.8%) |
Baseline Disease Characteristics
Table 13| Characteristic | SAKURA-1 DAXXIFY (N=201) | SAKURA-2 DAXXIFY (N=204) | SAKURA-1 Placebo (N=102) | SAKURA-2 Placebo (N=102) |
|---|---|---|---|---|
| Prior BoNT type A use, n (%) | 92 (45.8%) | 121 (59.3%) | 45 (44.1%) | 60 (58.8%) |
| IGA-FWS Moderate at baseline | 123 (61.2%) | 129 (63.2%) | 66 (64.7%) | 67 (65.7%) |
| IGA-FWS Severe at baseline | 78 (38.8%) | 75 (36.8%) | 36 (35.3%) | 35 (34.3%) |
| PFWS Moderate at baseline | 120 (59.7%) | 106 (52.0%) | 64 (62.7%) | 49 (48.0%) |
| PFWS Severe at baseline | 81 (40.3%) | 98 (48.0%) | 38 (37.3%) | 53 (52.0%) |
Efficacy Scale Definitions
Table 9| Score | Descriptor | Definition |
|---|---|---|
| 0 — None | No wrinkles | No visible glabellar wrinkles at maximum frown |
| 1 — Mild | Very shallow wrinkles | Barely perceptible wrinkles at maximum frown |
| 2 — Moderate | Moderate wrinkles | Moderate visible wrinkles at maximum frown |
| 3 — Severe | Deep and furrowed wrinkles | Deep, furrowed glabellar wrinkles at maximum frown |
Source: BLA 761127 Multi-disciplinary Review, Ref ID 5041522. IGA-FWS = Investigator Global Assessment-Frown Wrinkle Severity; PFWS = Patient Frown Wrinkle Severity; BoNT = botulinum neurotoxin. Demographics generally balanced across treatment groups. Higher proportion of Hispanic subjects in SAKURA-1 vs SAKURA-2 noted (23% vs 9%); consistent efficacy across subgroups confirmed.
Clinical Efficacy
Section 14.1 (Label) / Table 14, 20 (Medical Review)Primary Efficacy — 2-Point Composite Response at Week 4
ITT Population (as-randomized); Cochran-Mantel-HaenszelTreatment success: score ≤1 (none/mild) on both IGA-FWS and PFWS AND ≥2-grade improvement from baseline on both scales at Week 4. Zero placebo responders.
Week 4 — 2-Point Composite Treatment Success (Primary)
IGA-FWS ≤1 (Investigator)
PFWS ≤1 (Patient-Reported)
Identical design to SAKURA-1. Note: one subject randomized to placebo was inadvertently treated with DAXXIFY; included in placebo arm (as-randomized) for efficacy analysis. As-treated analysis: 74.2% vs 0%.
Week 4 — 2-Point Composite Treatment Success (Primary)
IGA-FWS ≤1 (Investigator)
PFWS ≤1 (Patient-Reported)
Efficacy Summary Table
Table 14 and 20, BLA 761127 Medical Review (Label Table 4)| Endpoint | SAKURA-1 DAXXIFY (N=201) | SAKURA-1 Placebo (N=102) | SAKURA-1 Δ (95% CI) | SAKURA-2 DAXXIFY (N=204) | SAKURA-2 Placebo (N=102) | SAKURA-2 Δ (95% CI) |
|---|---|---|---|---|---|---|
| Treatment Success Wk 4 (2-pt composite IGA + PFWS) | 148 (73.6%) | 0 (0%) | +74.2% (68.2–80.2%) p<0.0001 | 151 (74.0%) | 1 (1.0%) | +72.9% (66.6–79.1%) p<0.0001 |
| IGA-FWS ≤1 Wk 4 (Investigator only) | 176 (87.6%) | 1 (1.0%) | — | 187 (91.7%) | 3 (2.9%) | — |
| PFWS ≤1 Wk 4 (Patient only) | 155 (77.1%) | 0 (0%) | — | 156 (76.5%) | 1 (1.0%) | — |
| All secondary endpoints (IGA-FWS ≤1 and IGA+PFWS ≤1 at Wks 2–24) | All pre-specified secondary endpoints statistically significant in sequential testing (Weeks 2, 4, 8, 12, 16, 20, 24) in both trials | |||||
Subgroup Analysis — Primary Endpoint at Week 4
Table 19| Subgroup | SAKURA-1 DAXXIFY | SAKURA-1 Placebo | SAKURA-2 DAXXIFY | SAKURA-2 Placebo |
|---|---|---|---|---|
| Age 18–45 years | 50/58 (86%) | 0/32 (0%) | 49/62 (79%) | 1/30 (3%) |
| Age 46–55 years | 45/68 (66%) | 0/41 (0%) | 70/91 (77%) | 0/42 (0%) |
| Age 56–75 years | 53/75 (71%) | 0/29 (0%) | 32/51 (63%) | 0/30 (0%) |
| Female | 134/174 (77%) | 0/88 (0%) | 136/183 (74%) | 1/87 (1%) |
| Male | 14/27 (52%) | 0/14 (0%) | 15/21 (71%) | 0/15 (0%) |
| White | 126/173 (73%) | 0/81 (0%) | 133/180 (74%) | 1/92 (1%) |
| Black / African American | 8/10 (80%) | 0/8 (0%) | 8/9 (89%) | 0/3 (0%) |
| Asian | 6/7 (86%) | 0/2 (0%) | 7/11 (64%) | 0/5 (0%) |
| Hispanic or Latino | 39/47 (83%) | 0/25 (0%) | 15/19 (79%) | 1/10 (10%) |
Source: FDA label BLA 761127, Ref ID 5042005 (Table 4, Figure 2) and BLA 761127 Multi-disciplinary Review, Ref ID 5041522 (Tables 14, 20). ITT = intent-to-treat. Treatment success defined as IGA-FWS and PFWS score ≤1 (none or mild) AND ≥2-grade improvement from baseline on both scales. Composite “treatment success” endpoint is stringent — individual IGA-FWS response rates were 10–15% higher than composite rates. All secondary endpoints met in both trials using sequential multiplicity control.
Safety Profile
Section 6.1 / 6.2 (Label) / Tables 27–34 (Medical Review)Most Common Adverse Reactions ≥1% (Pooled SAKURA-1 + SAKURA-2 — Label Table 3)
N=406 DAXXIFY, N=203 Placebo| Adverse Reaction | DAXXIFY N=406 | Placebo N=203 | Notes |
|---|---|---|---|
| Headache | 26 (6%) | 4 (2%) | Most common ADR; pooled data (9.4% in medical review broader TEAE analysis). Includes tension headache. |
| Eyelid Ptosis | 9 (2%) | 0 (0%) | Local spread; 1% in SAKURA-OLS repeat-dose. Risk reduction: avoid injecting <1 cm above superior orbital rim. |
| Facial Paresis | 5 (1%) | 0 (0%) | Includes facial asymmetry; also 1.2% in SAKURA-OLS. Incidence did not increase with multiple retreatments. |
| Injection Site Reactions | 6% (pooled) | 6% (pooled) | Rates similar between DAXXIFY and placebo (not listed in label Table 3 as excess ADR). ISR components: pain 3.7%, erythema 1.2%, oedema 1.2%. Most mild and transient. |
TEAE Overview — SAKURA-1 + SAKURA-2 (Medical Review Table 27)
| TEAE Category | DAXXIFY N=406 | Placebo N=203 |
|---|---|---|
| Any TEAE — Mild | 208 (51.2%) | 51 (25.1%) |
| Any TEAE — Moderate | 56 (13.8%) | 16 (7.9%) |
| Any TEAE — Severe | 5 (1.2%) | 0 (0%) |
| Headache (incl. tension) | 38 (9.4%) | 5 (2.5%) |
| Upper respiratory tract infection | 28 (6.9%) | 10 (4.9%) |
| Injection site reaction (all types) | 25 (6.2%) | 13 (6.4%) |
| Eyelid ptosis | 9 (2.2%) | 0 (0%) |
| Influenza | 6 (1.5%) | 2 (1.0%) |
| Urinary tract infection | 5 (1.2%) | 0 (0%) |
| Facial paresis / asymmetry | 5 (1.2%) | 0 (0%) |
| SAEs (treatment-related) | 0 | 0 |
| Deaths | 1 death in SAKURA-OLS — not considered treatment-related | |
Repeat-Dose Safety (SAKURA-OLS, N=2691)
Warnings & Precautions
Section 5- Spread of Toxin Effect (5.1) — BOXED WARNING: Toxin effects may spread from injection site; symptoms hours to weeks post-injection including dysphagia, respiratory difficulties, generalized weakness. Deaths reported (other botulinum products). Not approved for spasticity or any condition other than glabellar lines.
- Non-Interchangeability (5.2): Units of DAXXIFY are NOT interchangeable with BOTOX, DYSPORT, XEOMIN, MYOBLOC, or JEUVEAU. Cannot compare or convert Units across products.
- Serious ADRs with Unapproved Use (5.3): Excessive weakness, dysphagia, aspiration pneumonia including fatal outcomes reported with unapproved botulinum toxin use. DAXXIFY not approved for any condition other than glabellar lines.
- Hypersensitivity Reactions (5.4): Anaphylaxis, serum sickness, urticaria, soft tissue edema, dyspnea reported with BoNT products. Discontinue and treat immediately.
- Cardiovascular System (5.5): Arrhythmia and MI including fatal outcomes reported with BoNT products. Use caution in patients with pre-existing cardiovascular disease.
- Pre-Existing Neuromuscular Disorders (5.6): Monitor patients with peripheral motor neuropathic diseases, ALS, myasthenia gravis, or Lambert-Eaton syndrome — at increased risk of clinically significant effects including severe dysphagia and respiratory compromise.
- Dysphagia and Breathing Difficulties (5.7): Can occur hours to weeks post-injection. Deaths as complication of severe dysphagia reported with BoNT products. Aspiration risk in patients with pre-existing swallowing compromise.
- Ophthalmic Adverse Reactions (5.9): Dry eye, reduced blinking, corneal disorders reported. If persistent eye symptoms: refer to ophthalmologist.
Immunogenicity
Section 6.2Source: FDA label BLA 761127, Ref ID 5042005; BLA 761127 Medical Review, Ref ID 5041522. AE profile considered similar to other licensed BoNT-A products for the same indication. No new safety signals identified. SAEs in SAKURA-OLS (N=29 subjects, 31 events) were predominantly unrelated to treatment. Reviewer concluded safety database of 2994 subjects (2839 at 40 U) was adequate and exceeded ICH E1A recommendations.
Pharmacology & Pharmacokinetics
Section 12 / LabelMechanism of Action
Section 12.1DAXXIFY blocks cholinergic transmission at the neuromuscular junction by inhibiting the release of acetylcholine (ACh). The mechanism proceeds in sequential steps: (1) binding of the toxin heavy chain to receptors on the presynaptic nerve terminal; (2) receptor-mediated endocytosis and internalization into the nerve terminal; (3) translocation of the light chain into the neuronal cytosol; (4) endopeptidase cleavage of SNAP25 (synaptosomal-associated protein 25 kDa) — a SNARE protein essential for synaptic vesicle docking and fusion. Cleavage of SNAP25 prevents the docking of ACh-containing vesicles to the plasma membrane, blocking exocytosis and neurotransmitter release, producing a dose-dependent decrease in muscle contractile function. Recovery of neuromuscular activity is gradual and results from two mechanisms: degradation of the neurotoxin light chain in the neurons, and formation of axonal sprouts leading to muscle reinnervation and slow reversal of the pharmacological effects.
Pharmacokinetics Summary
Section 12.3| PK Parameter | Details |
|---|---|
| Systemic detection | Cannot be detected in peripheral blood following IM injection at 40 Units using currently available analytical technology. No quantifiable systemic exposure data available for the approved indication. |
| Absorption | Locally distributed from IM injection site. Systemic absorption minimal or undetectable at therapeutic doses. Toxin undergoes retrograde axonal transport after receptor binding at NMJ. |
| Distribution | Local diffusion from injection site. The extent of diffusion is dose- and volume-dependent — higher doses or larger injection volumes increase risk of spread to adjacent muscles (local spread). |
| Metabolism | Degraded by normal protein catabolism. Light chain undergoes intraneuronal proteolysis. Heavy chain and accessory proteins cleared locally. |
| Elimination | Recovery of neuromuscular function via axonal sprouting/reinnervation; not via classical drug elimination. Duration of effect in glabellar muscles: clinically effective for approximately 24 weeks (median, composite endpoint). |
| N-CAM (neural cell adhesion molecule) | N-CAM expression in denervated motor end plates and axonal sprouts serves as a molecular marker for recovery — label section describes return to baseline N-CAM indicating complete reinnervation. |
| Drug-drug interactions | No formal DDI studies conducted. Theoretical potentiation expected with aminoglycosides (block presynaptic ACh release), anticholinergic agents, other BoNT products, and muscle relaxants. No pharmacokinetic DDI possible given undetectable systemic levels. |
Source: FDA label BLA 761127, Ref ID 5042005 (Section 12). No clinical pharmacokinetic data from human studies available for daxibotulinumtoxinA at the approved 40-Unit dose. This is consistent with all other approved botulinum toxin products where systemic concentrations are below the limits of detection of currently available assays. Product description per Section 11: sterile, preservative-free, white to off-white lyophilized powder in single-dose vials (50 U or 100 U) for intramuscular use after reconstitution with preservative-free 0.9% NaCl. U.S. License Number 2101.
Dosing, Administration & Contraindications
Section 2, 4, 5 / LabelReconstitution & Administration
Section 2.3 / 2.4Reconstitution Steps
- Slowly inject preservative-free 0.9% NaCl into vial (0.6 mL into 50 U vial; 1.2 mL into 100 U vial). Discard vial if vacuum does not pull diluent in.
- Gently rotate vial to mix. Do not shake. Reconstituted solution should be clear to slightly opalescent, colorless, free of particulate matter.
- Inspect visually before use. Do not use if cloudy, discolored, or contains particles.
- Use within 72 hours of reconstitution. Store unused reconstituted product at 2°C–8°C, protected from light. Do not freeze reconstituted product.
- Single patient, single session use only. Discard remaining solution immediately after injection.
Injection Technique
- Assess upper eyelid margin and levator palpebrae superioris function before injection. Evaluate levator function and frontalis compensation manually.
- Clean vial stopper with alcohol swab. Withdraw ≥0.5 mL with sterile syringe. Replace with 30–33 gauge sterile needle for injection.
- Expel air bubbles. Apply finger pressure on superior medial orbital rim.
- Inject 8 Units (0.1 mL) into each of 5 sites: medial corrugator (×2), lateral corrugator (×2), procerus (×1).
- Ptosis prevention: Do not inject <1 cm above superior orbital rim. Avoid levator palpebrae superioris area. Use accurate volumes. Use steady, controlled injection technique.
Contraindications
Section 4- Hypersensitivity (4.1): Known hypersensitivity to any botulinum toxin preparation, DAXXIFY, or any component of the DAXXIFY formulation (active or inactive ingredients). Cross-reactive hypersensitivity with other BoNT products is a contraindication.
- Infection at Injection Site (4.2): Presence of infection at the proposed injection sites.
Special Populations
Section 8| Population | Guidance |
|---|---|
| Pregnancy (8.1) | No human data. Animal IM studies: fetal growth effects (decreased weight and skeletal ossification) at doses ~40× MRHD (30 Units/kg in rats); embryofetal NOAEL 10 Units/kg (15× MRHD). No embryofetal toxicity in rabbits at up to MRHD (~0.48 Units/kg/day). No formal recommendation; weigh risks vs benefits. |
| Lactation (8.2) | No data on presence in human or animal milk, effects on breastfed infant, or milk production. Consider developmental/health benefits of breastfeeding alongside maternal clinical need. |
| Pediatric Use (8.4) | Safety and effectiveness in patients <18 years of age not established. DAXXIFY not approved for pediatric use. |
| Geriatric Use (8.5) | 36/406 subjects in placebo-controlled trials were ≥65 years. No increase in treatment-related AEs in patients >65 years. Insufficient numbers to determine whether elderly respond differently; caution advised for subgroup analyses showing lower response rates in 56–75 year age group (63–71% vs ~80–86% in 18–45 year group). |
| Cardiovascular disease | Use with caution. Arrhythmia and MI (some fatal) reported with BoNT products (Section 5.5). |
| Neuromuscular disorders | ALS, myasthenia gravis, Lambert-Eaton syndrome: monitor for exacerbated neuromuscular compromise. Peripheral motor neuropathic disease: increased risk of severe dysphagia and respiratory compromise. |
Storage & Supply
Section 16| Item | Detail |
|---|---|
| Unopened vials | Store at room temperature 20°C–25°C (68°F–77°F) OR refrigerated 2°C–8°C (36°F–46°F) in original carton, protected from light |
| After reconstitution | Use within 72 hours; store at 2°C–8°C, protected from light; do not freeze reconstituted product |
| 50 Unit vial NDC | 72960-111-01 (carton containing 1 vial) |
| 100 Unit vial NDC | 72960-112-01 (carton containing 1 vial) |
| Manufacturer | Revance Therapeutics, Inc., Newark, CA 94560. U.S. License Number 2101. |
| Drug interactions | No formal studies. Co-administer with caution: aminoglycosides, anticholinergic drugs, other BoNT neurotoxin products, muscle relaxants — all may potentiate effects |
Source: FDA label BLA 761127, Ref ID 5042005, Revised 9/2022. PI761127-0.8. Contact Revance Therapeutics at 1-877-373-8669 for adverse event reporting. FDA MedWatch: 1-800-FDA-1088.
Regulatory History
BLA 761127 — Biologics License ApplicationRegulatory Timeline
Regulatory Submission Summary
| Aspect | Detail |
|---|---|
| Application number | BLA 761127 (Orig1s000) |
| Application type | Biologic License Application (BLA) under 351(a) PHS Act; Standard Review |
| Applicant | Revance Therapeutics, Inc., Newark, CA 94560 |
| Submission / receipt date | November 24–25, 2019 |
| Original PDUFA goal | November 25, 2020 |
| CR letter date | October 14, 2021 (manufacturing only — no clinical deficiencies) |
| Resubmission date | March 8, 2022 |
| Review completion | September 6, 2022 |
| Approval date | September 8, 2022 |
| Pivotal trials | SAKURA-1 (Study 1620301; NCT data not specified in review) and SAKURA-2 (Study 1620302) — identical Phase 3 RCTs; N=609 combined |
| Supporting safety study | SAKURA-OLS (Study 1620303) — 84-week open-label repeat-dose study; N=2691 subjects, up to 3 treatment cycles |
| Total safety database | 3,139 subjects enrolled; 2,994 exposed to DAXXIFY at any dose; 2,839 at 40 Units (to-be-marketed dose) |
| Primary endpoint accepted | 2-point composite response at Week 4 (IGA-FWS + PFWS ≤1 AND ≥2-grade improvement on both scales) — FDA-endorsed stringent composite endpoint |
| Reference IDs | 5042005 (label, 9/2022) / 5041522 (multi-disciplinary review) / 5041615 (clinical review memorandum, resubmission) |
| U.S. License Number | 2101 |
Source: BLA 761127 label, Ref ID 5042005, Revised 9/2022; BLA 761127 Multi-disciplinary Review, Ref ID 5041522; BLA 761127 Clinical Review Memorandum (Resubmission), Ref ID 5041615, signed September 6, 2022. Reviewed under Division of Dermatology and Dentistry (DDD), Office of Immunology and Inflammation (OII), CDER, FDA. Team Lead: Tong Li-Masters MD, PhD, DDD. Project Manager: Kimberle Searcy.