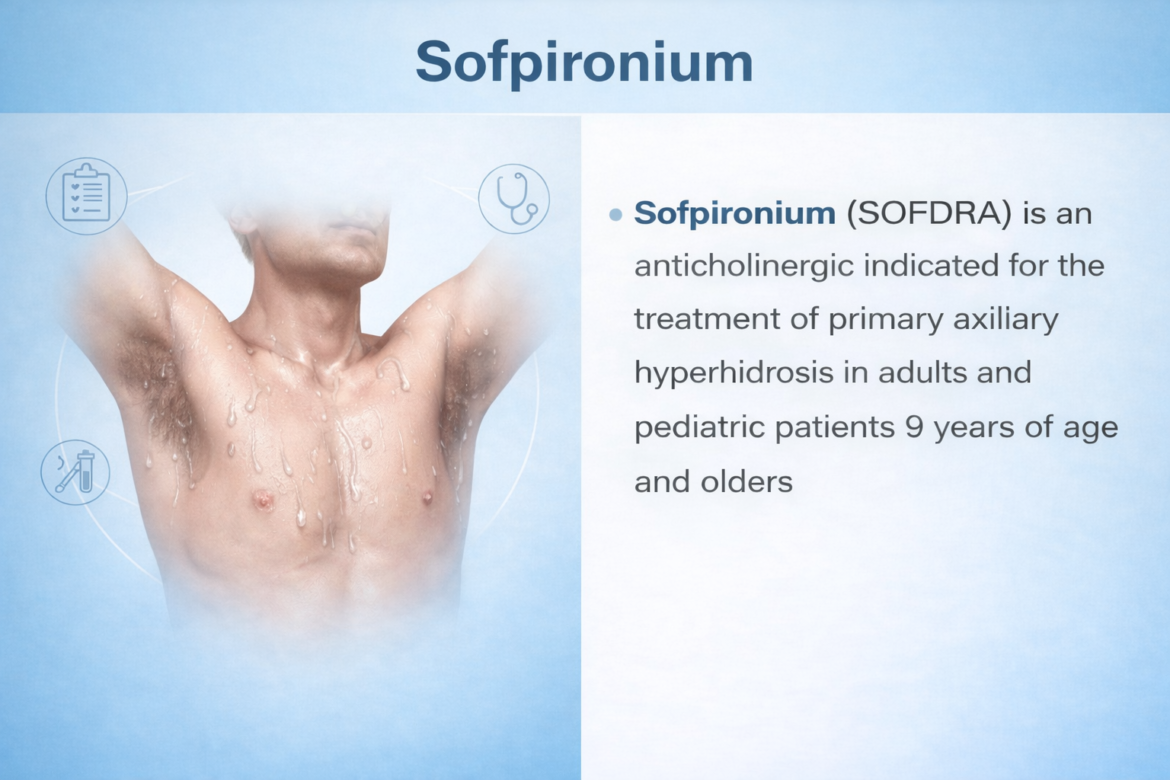
Sofpironium
Sofpironium bromide is a new molecular entity and small molecule quaternary ammonium analogue of the anticholinergic glycopyrrolate. It reduces sweat production through selective, competitive inhibition of muscarinic receptor type 3 (M3) — the predominant receptor in axillary eccrine glands. Applied once daily at bedtime via a metered pump applicator, it was studied in adults and pediatric patients ≥9 years with primary axillary hyperhidrosis. NDA 217347 was submitted September 2022 and received a Complete Response in September 2023 due to human factors deficiencies; the clinical discipline recommended approval following a Class 2 resubmission reviewed in June 2024.
Drug Overview
NDA 217347Drug Class & Chemistry
| Property | Detail |
|---|---|
| Pharmacologic class | Anticholinergic; muscarinic receptor M3 antagonist; quaternary ammonium analogue of glycopyrrolate |
| Code name | BBI-4000 |
| Molecular type | New molecular entity (NME); small molecule |
| Formulation | Gel, 15%; clear to slightly translucent, colorless to pale yellow; metered pump container with applicator cap |
| Dosing device | Metered pump bottle with applicator cap over the dispenser and a covering cap; one pump actuation per underarm per day |
| Sponsor | Botanix Pharmaceuticals |
| Japan approval | Sofpironium bromide gel 5% approved for marketing in Japan (tradename not specified in review) |
Mechanism Detail
Source: NDA 217347 Multi-disciplinary Review, Ref ID 5249224; Class 2 Resubmission Review, Ref ID 5398431. Review date: June 14, 2024.
Baseline Characteristics — Pivotal Trials
BBI-4000-CL-301 & 302 (ITT)Table 17 — Baseline Demographics (ITT)
| Characteristic | Study 301 SB Gel 15% N=173 | Study 301 Vehicle N=177 | Study 302 SB Gel 15% N=180 | Study 302 Vehicle N=171 |
|---|---|---|---|---|
| Age — Mean (SD), years | 32.9 (11.6) | 32.4 (10.9) | 32.1 (12.2) | 31.7 (11.1) |
| Age — Range | 11, 64 | 14, 71 | 10, 76 | 11, 69 |
| Age 9–11 years, n (%) | 1 (1%) | 0 (0%) | 2 (1%) | 2 (1%) |
| Age 12–17 years, n (%) | 14 (8%) | 12 (7%) | 13 (7%) | 8 (5%) |
| Age 18–64 years, n (%) | 158 (91%) | 163 (92%) | 161 (89%) | 158 (92%) |
| Age ≥65 years, n (%) | 0 (0%) | 2 (1%) | 4 (2%) | 3 (2%) |
| Female, n (%) | 98 (57%) | 99 (56%) | 92 (51%) | 103 (60%) |
| Male, n (%) | 75 (43%) | 78 (44%) | 88 (49%) | 68 (40%) |
| White, n (%) | 140 (81%) | 130 (73%) | 141 (78%) | 133 (78%) |
| Black or African American, n (%) | 31 (18%) | 43 (24%) | 32 (18%) | 34 (20%) |
| Asian, n (%) | 1 (1%) | 3 (2%) | 4 (2%) | 1 (1%) |
| American Indian or Alaska Native, n (%) | 0 (0%) | 0 (0%) | 2 (1%) | 1 (1%) |
| Unknown / not reported, n (%) | 1 (1%) | 1 (1%) | 1 (1%) | 2 (1%) |
| Hispanic or Latino, n (%) | 61 (35%) | 60 (34%) | 48 (27%) | 45 (26%) |
| Not Hispanic or Latino, n (%) | 111 (64%) | 117 (66%) | 132 (73%) | 124 (73%) |
| Not reported, n (%) | 1 (1%) | 0 (0%) | 0 (0%) | 2 (1%) |
Note: ITT population includes duplicate subject enrollments. Race reflects predominant race if multiple races selected. Source: CSR 301 (page 58) and CSR 302 (page 60); reproduced by statistical reviewer using adsl.xpt.
Table 18 — Baseline Disease Characteristics (ITT)
| Characteristic | Study 301 SB Gel 15% N=173 | Study 301 Vehicle N=177 | Study 302 SB Gel 15% N=180 | Study 302 Vehicle N=171 |
|---|---|---|---|---|
| HDSM-Ax-7 — Mean (SD) | 3.5 (0.3) | 3.5 (0.3) | 3.6 (0.3) | 3.6 (0.3) |
| HDSM-Ax-7 — Median | 3.4 | 3.6 | 3.6 | 3.6 |
| HDSM-Ax-7 — Range | 3.0, 4.0 | 2.9, 4.0 | 3.0, 4.0 | 3.0, 4.0 |
| GSP — Mean (SD), mg | 296.2 (257.1) | 298.5 (223.4) | 313.8 (283.7) | 308.4 (267.2) |
| GSP — Median, mg | 214.1 | 228.6 | 207.7 | 231.1 |
| GSP — Range, mg | 38.4, 1512.1 | 20.4, 1523.8 | 38.6, 1773.6 | 8.9, 2356.6 |
| Duration of Symptoms — Mean (SD), months | 179.3 (115.2) | 184.4 (116.9) | 202.6 (131.9) | 194.4 (117.3) |
| Duration of Symptoms — Median, months | 156.3 | 157.2 | 193.6 | 172.4 |
| Duration of Symptoms — Range, months | 6.5, 564.7 | 19.9, 588.6 | 9.1, 712.0 | 16.5, 495.4 |
HDSM-Ax-7 = Hyperhidrosis Disease Severity Measure-Axillary-7-item scale; Baseline/Day 1 (Visit 4) assessment. GSP = Gravimetric Sweat Production; baseline value is the median of observations on Visits 2, 3, and 4 (Visits 2 and 3 within 14 days prior to Baseline/Day 1). Source: Reviewer analysis using adhdsm.xpt, adgsp.xpt, and adsl.xpt.
Table 15 — Subject Disposition (Studies 301 and 302)
| Population / Disposition | Study 301 SB Gel 15% N=173 | Study 301 Vehicle N=177 | Study 302 SB Gel 15% N=180 | Study 302 Vehicle N=171 |
|---|---|---|---|---|
| ITT¹, n (%) | 173 (100%) | 177 (100%) | 180 (100%) | 171 (100%) |
| mITT², n (%) | 169 (98%) | 173 (98%) | 180 (100%) | 169 (99%) |
| Safety Population³, n (%) | 173 (100%) | 176 (99%) | 180 (100%) | 171 (100%) |
| Discontinued treatment, n (%) | 19 (11%) | 10 (6%) | 19 (11%) | 13 (8%) |
| Discontinued study, n (%) | 23 (13%) | 15 (8%) | 20 (11%) | 16 (9%) |
| — Adverse event | 5 (3%) | 0 (0%) | 9 (5%) | 0 (0%) |
| — Lost to follow-up | 10 (6%) | 8 (5%) | 4 (2%) | 8 (5%) |
| — Withdrawal by subject | 4 (2%) | 4 (2%) | 6 (3%) | 6 (4%) |
| — Protocol violation | 2 (1%) | 3 (2%) | 0 (0%) | 2 (1%) |
| — Pregnancy | 1 (1%) | 0 (0%) | 0 (0%) | 0 (0%) |
| — Physician decision | 0 (0%) | 0 (0%) | 1 (1%) | 0 (0%) |
| — Other | 1 (1%) | 0 (0%) | 0 (0%) | 0 (0%) |
¹ All randomized subjects (includes subjects with multiple randomizations). ² All subjects randomized only once. ³ Randomized and treated subjects (includes subjects with multiple randomizations). Note: All subjects who discontinued due to adverse events were on the SB gel arm. Four subjects were randomized multiple times across both studies and are excluded from mITT. Source: CSR 301 (page 52) and CSR 302 (page 54); reproduced by statistical reviewer using adsl.xpt.
Study Design Summary
| Parameter | Detail |
|---|---|
| Design | Multicenter, randomized, double-blind, vehicle-controlled, parallel-group Phase 3 pivotal efficacy and safety study |
| Randomization | 1:1 (SB gel 15%:vehicle); approximately 350 subjects per study |
| Treatment duration | 42 days (6 weeks); once-daily application at bedtime to each axilla; planned mean duration ~42 days |
| Key inclusion criteria | Age ≥9 years; primary axillary hyperhidrosis; HDSM-Ax-7 score 3–4 at screening and baseline; gravimetric sweat production ≥50 mg/axilla and ≥150 mg combined (both axillae) in 5 minutes; symptoms ≥6 months’ duration |
| Co-primary endpoints | (1) Proportion of subjects ≥12 years with ≥2-point improvement in HDSM-Ax-7 score from baseline to end of treatment (EOT); (2) Change from baseline to EOT in gravimetric sweat production (GSP; ranked analysis) |
| Sites | Study 301: 39 U.S. sites, 35 randomized subjects. Study 302: 42 U.S. sites, 33 randomized subjects |
| Trial identifiers | Study 301: BBI-4000-CL-301 (NCT03836287) | Study 302: BBI-4000-CL-302 (NCT03948646) |
| Pediatric note | Only 5 subjects aged 9 to <12 years enrolled across both studies; HDSM-Ax-7 Child Version used; results presented for ≥12 years (Adult Version) per FDA recommendation |
Source: NDA 217347 Multi-disciplinary Review, Ref ID 5249224, Tables 15, 17, and 18 (Section 8). ITT = all randomized subjects. HDSM-Ax-7 = Hyperhidrosis Disease Severity Measure-Axillary-7-item scale. GSP = Gravimetric Sweat Production.
Clinical Efficacy
Studies BBI-4000-CL-301 & 302Co-Primary Endpoint 1 — ≥2-Point HDSM-Ax-7 Improvement (≥12 years, ITT)
Co-Primary Endpoint 2 — Change from Baseline in Gravimetric Sweat Production (ITT)
Co-Primary Efficacy Summary Table
| Endpoint | Study 301 SB Gel N=173 | Study 301 Vehicle N=177 | Study 302 SB Gel N=180 | Study 302 Vehicle N=171 |
|---|---|---|---|---|
| ≥2-pt HDSM-Ax-7 improvement (≥12 yrs) | 50.4% | 32.1% | 63.3% | 47.1% |
| Treatment difference (95% CI) | 18.3% (8.0, 28.7) | 16.2% (5.4, 26.9) | ||
| p-value (HDSM-Ax-7) | 0.0005 | 0.0040 | ||
| Median change in GSP (mg) | −127.8 | −100.3 | −142.6 | −134.2 |
| p-value (GSP ranked analysis) | 0.0019 | 0.0296 | ||
Source: NDA 217347 Multi-disciplinary Review, Ref ID 5249224, Table 1 and Tables 22–29. ITT population; multiple imputation. GSP ranked analysis per protocol (non-normality confirmed). HDSM-Ax-7 = Hyperhidrosis Disease Severity Measure-Axillary-7-item scale; GSP = Gravimetric Sweat Production.
Safety & Adverse Drug Reactions
Studies 301, 302, & 303Key Safety Considerations
Warnings & Precautions
- Anticholinergic effects — dilated pupils, blurry vision, dry eye, dry mouth, constipation, urinary hesitation. Monitor patients, particularly those with conditions predisposing to urinary retention.
- Local skin reactions — application site pain, redness, dermatitis, pruritus reported >1% more frequently with SB gel than vehicle. Discontinue if severe local reactions occur.
- Human Factors — original HF validation study identified critical use errors with metered pump applicator. Revised user interface and IFU implemented following Complete Response; new HF validation study submitted April 2024.
Use in Specific Populations
Source: NDA 217347 Multi-disciplinary Review, Ref ID 5249224; Class 2 Resubmission Review, Ref ID 5398431. Safety population = randomized and treated subjects.
Dosing & Contraindications
Standard Dosing
- Apply sofpironium bromide gel 15% once daily at bedtime
- One pump actuation per underarm (axilla)
- Applied using the pump cap applicator onto each axilla
- Wash hands with soap and water immediately after use
- Applied topically; not for oral, ophthalmic, or other routes
Special Populations
- Pediatric (≥9 years): Proposed indication; studied at same dose as adults (one pump per axilla QD)
- Pediatric (<9 years): Not studied; not approved
- Geriatric: No specific dose adjustment stated; limited trial data in ≥65 years
- Renal/Hepatic impairment: Not reported in medical review — systemic exposure low with topical application
- Pregnancy/Lactation: Insufficient human data; use only if benefit justifies risk
Contraindications
- Not reported in label as specified in the medical review documents provided
Device / Application Instructions
- Metered pump container with applicator cap over dispenser and covering cap
- Applicator cap labeled with intended use/purpose (per revised IFU post-CR)
- IFU revised to reduce complexity and improve salience of key steps
- “Wash hands with soap and water immediately after use” placed on principal display panel of carton and container labeling
- Revised to address tilting/spilling use error identified in first HF validation study
Source: NDA 217347 Multi-disciplinary Review, Ref ID 5249224; Class 2 Resubmission Review, Ref ID 5398431.
Labeling
NDA 217347| Item | Detail |
|---|---|
| Product name | Sofdra (sofpironium bromide) gel, 15% |
| Dosage form | Gel; clear to slightly translucent, colorless to pale yellow |
| Concentration | 15% sofpironium bromide |
| Container | Metered pump container with applicator cap and covering cap |
| Applicant | Botanix Pharmaceuticals |
| Proprietary name review | Sofdra — conditionally approved tradename; DMEPA reviewed March 13, 2024; concluded acceptable June 11, 2024 |
| Storage | Not reported in label sections provided in the medical review |
| NDC | Not reported in the medical review documents provided |
Labeling History & IFU Revisions
- Original IFU identified as having too much information, certain information lacking salience, and being folded (contributing to missed steps) — per first HF validation study.
- Revised IFU submitted December 20, 2023 as part of Class 2 resubmission (SDN 24); updated draft PI, IFU, carton and container labeling with proprietary name included.
- Statement “wash hands with soap and water immediately after use” added to principal display panel of carton and container labeling per CR requirement.
- Applicator labeled with intended use/purpose to address users not understanding what the applicator was or how to use it.
Adverse Event Reporting
| Contact | Detail |
|---|---|
| Reference IDs | 5249224 (original cycle multi-disciplinary review) / 5398431 (Class 2 resubmission review, June 14, 2024) |
| Reviewing division | Division of Dermatology and Dentistry (DDD), Office of Immunology and Inflammation (OII), CDER |
| Clinical reviewer | Roselyn E. Epps, M.D., FAAP, FAAD; Clinical Team Leader: David Kettl, M.D. |
Source: NDA 217347 Class 2 Resubmission Review, Ref ID 5398431; NDA 217347 Multi-disciplinary Review, Ref ID 5249224.
Regulatory History
NDA 217347 — Standard ReviewRegulatory Timeline
Regulatory Submission Summary
| Aspect | Detail |
|---|---|
| Application number | NDA 217347 (Orig1s000) |
| Applicant | Botanix Pharmaceuticals |
| Submission / receipt date | September 23, 2022 |
| PDUFA goal date | September 23, 2023 |
| Review type | Standard Review; Original NDA; New Molecular Entity |
| Complete Response date | September 22, 2023 — HF deficiency only |
| Class 2 resubmission date | December 20, 2023 (SDN 24) |
| Pivotal trials | BBI-4000-CL-301 (NCT03836287) and BBI-4000-CL-302 (NCT03948646) |
| Co-primary endpoints met | Study 301: HDSM-Ax-7 50.4% vs 32.1% (p=0.0005); GSP ranked p=0.0019. Study 302: HDSM-Ax-7 63.3% vs 47.1% (p=0.0040); GSP ranked p=0.0296. All statistically significant. |
| Boxed Warning | Not reported in review as required |
| REMS | Not required |
| Priority Review / Breakthrough | Standard Review; no breakthrough or priority designation reported |
| Japan approval | Sofpironium bromide gel 5% approved in Japan; pharmacovigilance report submitted with resubmission — events consistent with approved labeling |
| Reference IDs | 5249224 (original cycle multi-disciplinary review) / 5398431 (Class 2 resubmission review, June 14, 2024) |
Source: NDA 217347 Class 2 Resubmission Review, Ref ID 5398431 (June 14, 2024); NDA 217347 Multi-disciplinary Review, Ref ID 5249224. Reviewed under Division of Dermatology and Dentistry (DDD), Office of Immunology and Inflammation (OII), CDER.