Bimekizumab
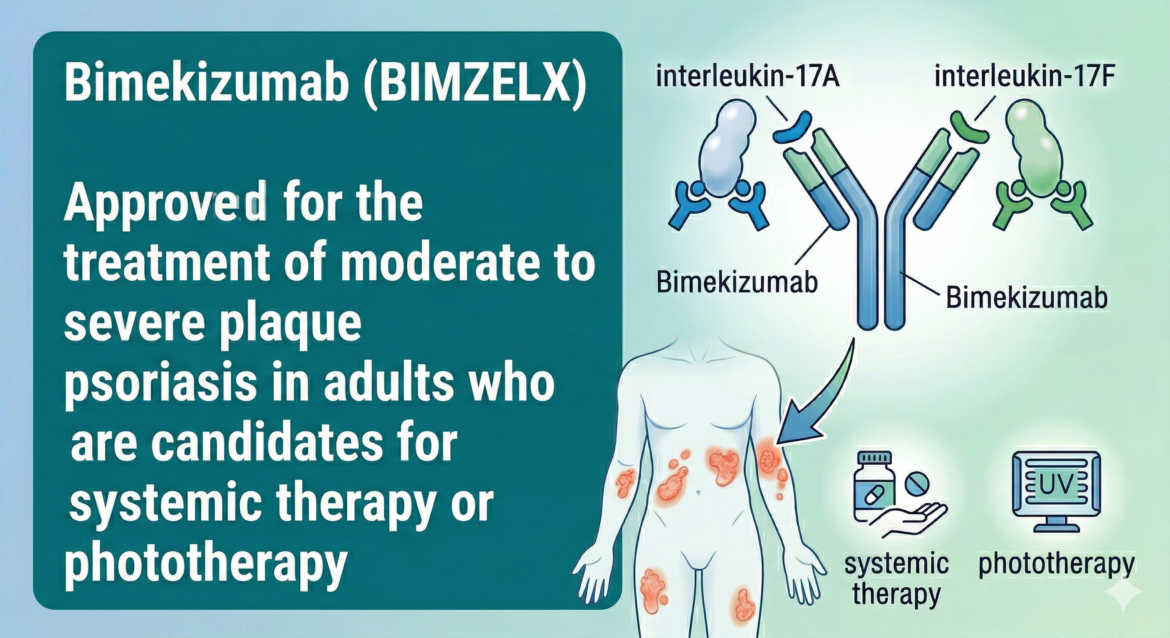
A humanized IgG1 monoclonal antibody that selectively inhibits both interleukin-17A (IL-17A) and interleukin-17F (IL-17F) — the first dual IL-17A/F inhibitor approved by the FDA. Indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy.
Drug Overview
Mechanism of Action
Bimekizumab-bkzx is a humanized IgG1/κ monoclonal antibody with two identical antigen-binding regions that selectively bind to human interleukin-17A (IL-17A), interleukin-17F (IL-17F), and interleukin-17AF heterodimer cytokines, inhibiting their interaction with the IL-17 receptor complex.
IL-17A and IL-17F are naturally occurring cytokines involved in normal inflammatory and immune responses. Elevated levels of both are found in lesional psoriatic skin. Unlike IL-17A-only inhibitors (secukinumab, ixekizumab), bimekizumab uniquely neutralizes both IL-17A and IL-17F, which may contribute to its high rates of complete skin clearance. Bimekizumab inhibits the release of proinflammatory cytokines and chemokines.
Competitive Landscape
| Drug | Target | Route | Approval | PASI 90 (vs placebo) |
|---|---|---|---|---|
| Secukinumab (COSENTYX) | IL-17A | SC | 2015 | ~75% vs 3–4% |
| Ixekizumab (TALTZ) | IL-17A | SC | 2016 | ~70% vs 3% |
| Brodalumab (SILIQ) | IL-17RA | SC | 2017 | ~83% vs 3% |
| Guselkumab (TREMFYA) | IL-23 | SC | 2017 | ~73% vs 3% |
| Risankizumab (SKYRIZI) | IL-23p19 | SC | 2019 | ~72% vs 2% |
| Bimekizumab (BIMZELX) ▶ | IL-17A + IL-17F | SC | Oct 2023 | 85–91% vs 1–5% |
Key Drug Information
| Parameter | Detail |
|---|---|
| Proprietary Name | BIMZELX® |
| INN / Generic Name | Bimekizumab-bkzx |
| Code Name | UCB4940 (CDP4940) |
| Sponsor / Manufacturer | UCB, Inc. · 1950 Lake Park Drive, Smyrna, GA 30080 · US License No. 1736 |
| Pharmacologic Class | Humanized interleukin-17A and F antagonist (IgG1 monoclonal antibody) |
| Molecular Weight | ~150 kDa |
| Production | Recombinant DNA technology in Chinese Hamster Ovary (CHO) cells |
| Dosage Forms | 160 mg/mL in single-dose prefilled syringe or single-dose prefilled autoinjector (1 mL) |
| NDC | 50474-781-85 (autoinjector carton, 2×160 mg) · 50474-780-79 (prefilled syringe carton, 2×160 mg) |
| Inactive Ingredients | Glacial acetic acid, glycine, polysorbate 80, sodium acetate, Water for Injection USP (pH 5.1) |
| Storage | Refrigerated 2°C–8°C. May store at room temp ≤25°C for up to 30 days in original carton. Do not freeze or shake. |
| PI Revision Date | October 2023 |
Clinical Efficacy
at Week 16
Co-Primary Endpoint Results at Week 16
IGA 0 or 1 (“Clear” or “Almost Clear”) at Week 16
PASI 90 at Week 16
IGA 0 or 1 at Week 16
PASI 90 at Week 16
Secondary Endpoints at Week 16 (Trials Ps-1 & Ps-2)
| Endpoint | Ps-1 BIMZELX | Ps-1 Placebo | Ps-2 BIMZELX | Ps-2 Placebo |
|---|---|---|---|---|
| IGA 0 (“Clear”) | 59% | 0% | 70% | 1% |
| PASI 100 (Complete clearance) | 59% | 0% | 68% | 1% |
| PASI 75 at Week 4 | 77% | 2% | 76% | 1% |
| Scalp IGA 0/1 at Week 16 | 84% (240/285) | 15% (11/72) | 92% (286/310) | 7% (5/74) |
Efficacy Across Endpoints — Week 16 Visual Summary
Maintenance of Response — Week 56 (Trial Ps-2 Randomized Withdrawal)
Active-Comparator Head-to-Head Data
| Trial | Comparator | BIMZELX PASI 90 | Comparator PASI 90 | Significance |
|---|---|---|---|---|
| Ps-1 (NCT03370133) | Ustekinumab (45 mg or 90 mg) | 85% at Wk 16 | ~50% at Wk 16 | Superior |
| Ps-3 (NCT03412747) | Adalimumab (80 mg then 40 mg Q2W) | ~86% at Wk 16 | ~48% at Wk 16 | Superior |
| Ps-4 (PS0015) | Secukinumab 300 mg | ~86% at Wk 16 | ~70% at Wk 16 | Superior |
Safety & Adverse Drug Reactions
Warnings and Precautions
- Suicidal Ideation and Behavior (SI/B): May increase risk. Monitor for emergence or worsening of depression, suicidal ideation, or mood changes. Call National Suicide and Crisis Lifeline at 988 if changes occur.
- Serious Infections: May increase risk. Do not administer until active infection resolves.
- Tuberculosis: Evaluate before treatment. Avoid in active TB. Initiate treatment of latent TB prior to use.
- Liver Biochemical Abnormalities: Test liver enzymes at baseline and periodically. Permanently discontinue if causally associated combined transaminase/bilirubin elevations occur.
- Inflammatory Bowel Disease: Avoid in active IBD. Monitor for new or worsening IBD symptoms.
Adverse Reactions ≥1% — 16-Week Controlled Data (Trials Ps-1 & Ps-2)
| Adverse Reaction | BIMZELX 320 mg (N=670) | Placebo (N=169) |
|---|---|---|
| Upper Respiratory Tract Infections | 102 (15%) | 24 (14%) |
| Oral Candidiasis | 61 (9%) | 0 (0%) |
| Headache | 22 (3%) | 0 (0%) |
| Injection Site Reactions | 19 (3%) | 2 (1%) |
| Tinea Infections | 18 (3%) | 1 (1%) |
| Gastroenteritis | 12 (2%) | 0 (0%) |
| Herpes Simplex Infections | 9 (1%) | 0 (0%) |
| Acne | 8 (1%) | 0 (0%) |
| Folliculitis | 8 (1%) | 0 (0%) |
| Other Candida Infections | 7 (1%) | 1 (1%) |
| Fatigue | 7 (1%) | 0 (0%) |
Key Safety Signals
Long-Term Safety (Weeks 56–128, Open-Label Extension)
Through Week 128, no new adverse reactions beyond those identified in the initial 16-week period were identified — except for suicide attempts and completed suicide occurring in the OLE. Through Week 144 (Trial Ps-4, 691 subjects), no new adverse reactions were identified.
Use in Special Populations
| Population | Summary |
|---|---|
| Pregnancy | Insufficient human data. No adverse developmental effects in cynomolgus monkeys up to 38× MRHD (50 mg/kg/week SC). IgG crosses placenta — delay live vaccines in infants exposed in utero (consider ≥4 months after birth). Pregnancy registry: 1-877-311-8972. |
| Lactation | No data in human or animal milk. Endogenous IgG transferred in human milk. Benefits of breastfeeding should be considered alongside risks. |
| Pediatric | Safety and effectiveness not established in pediatric patients. |
| Geriatric (≥65 yr) | 153/1,789 subjects were ≥65 years; 18 were ≥75 years. No differences in safety or effectiveness observed, but numbers insufficient for definitive conclusions. |
| High Body Weight (≥120 kg) | Average plasma concentrations ~30% lower in subjects ≥120 kg vs <120 kg. Consider 320 mg Q4W (instead of Q8W) after Week 16 in patients ≥120 kg. |
Pharmacology & Pharmacokinetics
Mechanism of Action
Bimekizumab-bkzx selectively binds and neutralizes IL-17A, IL-17F, and the IL-17AF heterodimer, preventing their interaction with the IL-17 receptor complex. Both IL-17A and IL-17F are involved in psoriasis pathogenesis — they are elevated in psoriatic lesions and drive keratinocyte activation, neutrophil recruitment, and production of proinflammatory mediators. Dual neutralization of both cytokines may provide greater suppression of the psoriatic inflammatory cascade compared to IL-17A-only inhibitors.
Pharmacokinetic Parameters
Metabolism & Excretion
Bimekizumab-bkzx is expected to be degraded into small peptides by catabolic pathways, consistent with other IgG1 monoclonal antibodies. No dedicated hepatic or renal metabolism studies are required for large biologics of this class.
Drug Interactions
Pharmacodynamics
Elevated levels of IL-17A and IL-17F are found in lesional psoriatic skin. Bimekizumab exposure-response relationships to serum biomarkers (including IL-17A and IL-17F) and the time course of pharmacodynamic responses are not fully characterized.
Immune Response to Inactivated Vaccines: Healthy subjects who received a single 320 mg dose of BIMZELX 2 weeks prior to inactivated seasonal influenza vaccination had similar antibody responses compared to unvaccinated controls. Avoid live vaccines during BIMZELX treatment.
Body Weight Effect
In subjects weighing ≥120 kg, average plasma concentrations of bimekizumab are predicted to be at least 30% lower than in those weighing <120 kg (median 87 kg). This is why dosing guidance recommends considering 320 mg Q4W after Week 16 for patients ≥120 kg.
Dosing & Administration
Dosing Schedule
| Period | Dose | Frequency | Notes |
|---|---|---|---|
| Induction (Weeks 0–16) | 320 mg (2 × 160 mg SC) | Q4W: Weeks 0, 4, 8, 12, 16 | Two injections at different anatomical sites per dose |
| Maintenance (after Week 16) | 320 mg | Every 8 weeks (Q8W) | Standard maintenance dose |
| High body weight (≥120 kg) | 320 mg | Consider Q4W after Week 16 | Plasma concentrations ~30% lower in ≥120 kg patients |
| Missed dose | 320 mg | Administer as soon as possible | Resume regular schedule thereafter |
Pre-Treatment Evaluations Required
Administration Instructions
| Parameter | Instruction |
|---|---|
| Preparation | Remove from refrigerator 30–45 minutes before injection to reach room temperature (protects from light in original carton) |
| Injection sites | Thigh, abdomen (not within 2 inches of navel), or back of upper arm (HCP/caregiver only) |
| Avoid injecting into | Tender, bruised, red, hard, thick, scaly skin or areas affected by psoriasis |
| Site rotation | Different anatomical locations for each of the two 160 mg injections per dose; alternate sites each administration |
| Self-injection | Patients may self-inject after proper training in subcutaneous injection technique by a healthcare professional |
| Syringe/autoinjector disposal | Discard in FDA-cleared sharps container immediately after use. Do not reuse. |
Contraindications
Regulatory History
BLA 761151 — Key Facts
| Parameter | Detail |
|---|---|
| Application Number | BLA 761151 |
| Application Type | BLA (Resubmission after Complete Response) · Standard review |
| Sponsor | UCB, Inc. |
| Code Name | UCB4940 (CDP4940) |
| Original BLA Submission | July 15, 2020 |
| Complete Response Letter | May 12, 2022 (GMP deficiencies at UCB Braine facility) |
| BLA Resubmission Date | November 21, 2022 |
| PDUFA Goal Date | May 21, 2023 |
| Review Completion Date | October 16, 2023 |
| FDA Approval Date | October 2023 |
| Review Division | Division of Dermatology and Dentistry / Office of Immunology and Inflammation |
| Medical Review Ref ID | 5261174 (resubmission) · 4982337 (original) |
| PI Ref ID | 5262329 |
| EU Approval | August 2023 — European Commission |
Regulatory Timeline
FDA Review Assessment
Post-Marketing Requirements
| Requirement | Detail |
|---|---|
| Suicidal Ideation Pharmacovigilance | Ongoing monitoring of SI/B through post-marketing surveillance. Updated labeling and Medication Guide to communicate risk. |
| Pediatric Studies | Safety and efficacy not established in pediatric patients; data required per PREA. |
| Pregnancy Registry | OTIS AutoImmune Diseases Study — 1-877-311-8972 or mothertobaby.org/pregnancy-studies/ |
| Long-term Liver Safety | Continued monitoring and reporting of liver biochemical abnormalities per routine pharmacovigilance. |
For investigational and educational purposes only. Not for clinical decision-making.
© 2026 TrialistMD · All rights reserved