NAVEPEGRITIDE (YUVIWEL)
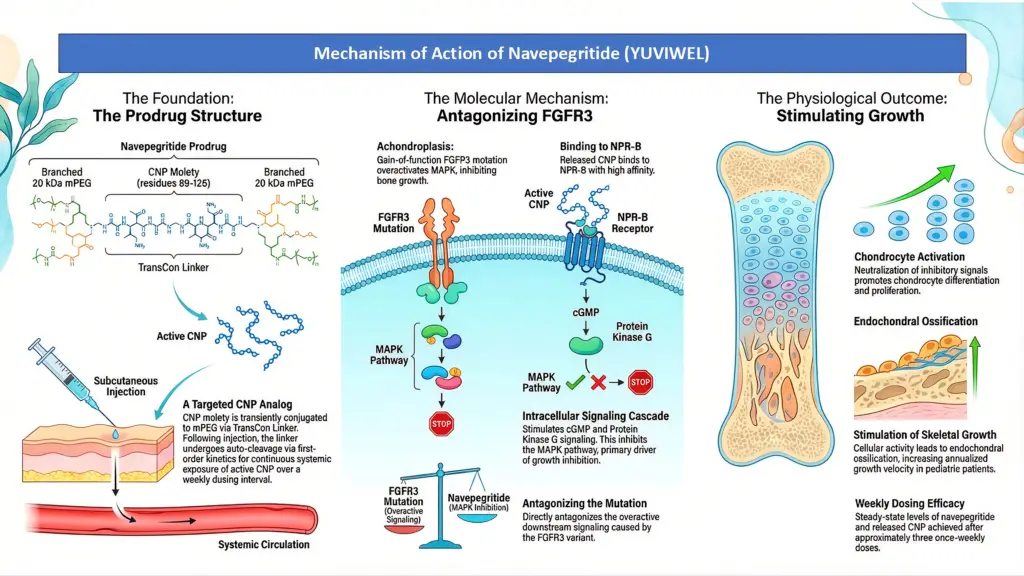
Navepegritide (YUVIWEL) is a once-weekly subcutaneous injection designed to enhance linear growth in pediatric patients aged two and older who have achondroplasia and open growth plates(ephiphyses). This C-type natriuretic peptide (CNP) analog works by stimulating bone growth and was granted accelerated approval based on its ability to improve growth velocity in clinical trials.
The medication is provided as a lyophilized powder that must be reconstituted with a supplied diluent before use, with specific weight-based dosages ranging from 1.3 mg to 5.5 mg.
Common side effects include vomiting, nausea, and injection-site reactions, while healthcare providers warn of a potential risk for transient low blood pressure. Usage is generally not recommended for those with moderate to severe renal impairment, and continuous monitoring of a child’s growth is required to determine when treatment should cease.
Yuviwel (navepegritide)
Drug Overview
Structured clinical and regulatory framing for quick interpretation at the point of review.
Core profile
Regulatory status
Pharmacokinetics
Approval-era PK interpretation based on the U.S. prescribing information, paired with an illustrative exposure profile.
Illustrative normalized exposure profile
Modeled from approval-label Tmax and half-life values. This curve is an inference for visual orientation, not a reproduced source figure.
Clinical Trial Database
Structured trial records designed for rapid reading, with expandable details, endpoint tables, and publication links.
Interactive Analytics Dashboard
Filter endpoints, compare trials, inspect subgroup effects, and export visual outputs for slide decks or research notes.
Endpoint comparison
Treatment, comparator, and filtered effect estimates across the active selection.
Trial trend view
Uses each study’s reusable trend series for dose-response or age-band effect visualization.
Forest plot
Subgroup or cohort-level point estimates with confidence intervals, tuned to the active trial and subgroup selection.
Sources
Primary-source references used to build the dashboard. Approval, PK, and trial values were taken from these materials and then normalized into the dataset layer.
Hi, this is a comment.
To get started with moderating, editing, and deleting comments, please visit the Comments screen in the dashboard.
Commenter avatars come from Gravatar.