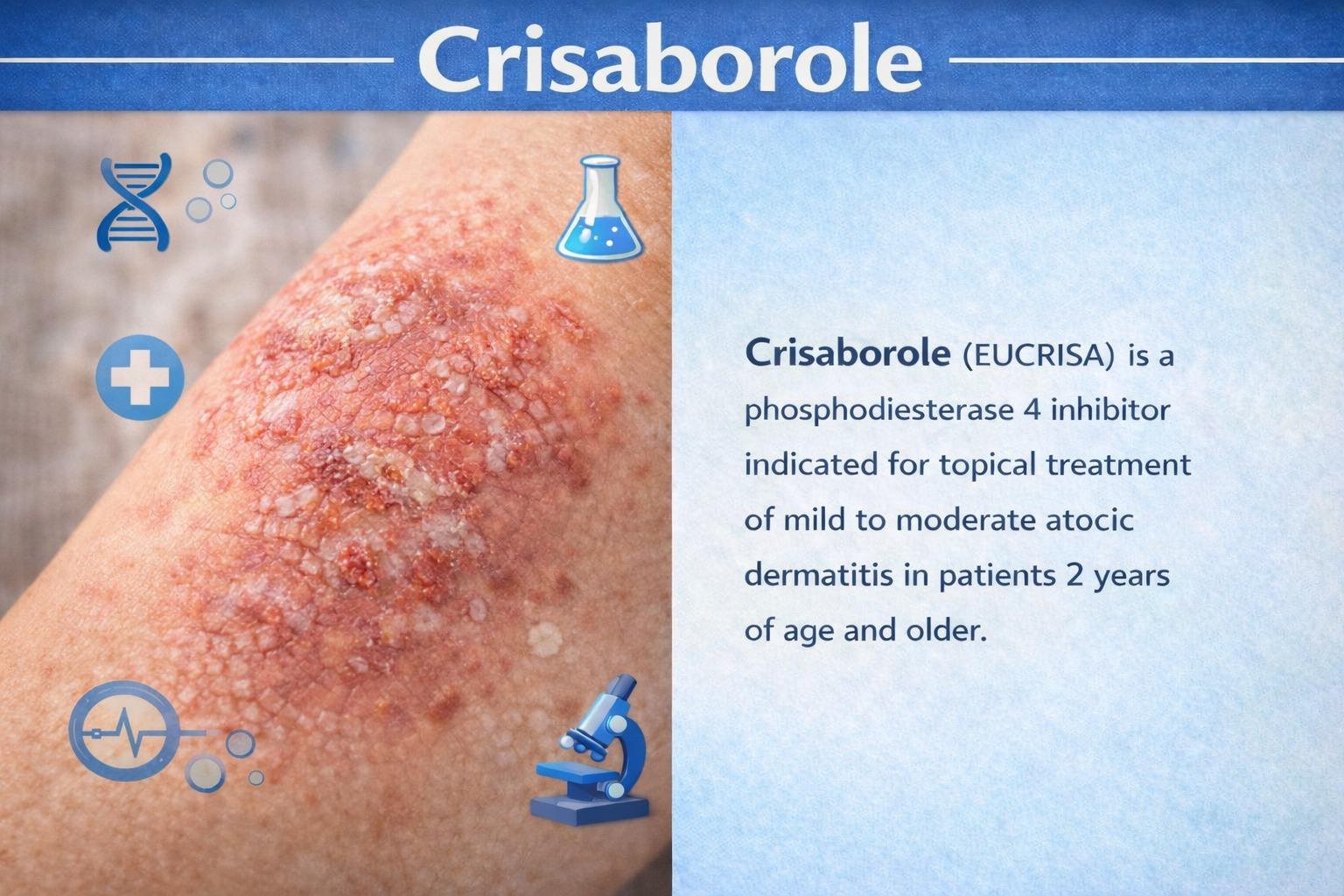
Indication
Mild–Moderate AD ≥2 yr
Topical treatment of mild to moderate atopic dermatitis in patients 2 years of age and older
Mechanism
PDE-4 Inhibitor
Boron-based benzoxaborole small molecule; topical non-steroidal
Dose & Schedule
Thin layer BID
Applied twice daily to affected areas; topical use only
Phase 3 Programme
2 trials
N=1,522 (ITT); plus long-term safety trial N=517
Mechanism of Action: Crisaborole is a phosphodiesterase 4 (PDE-4) inhibitor. PDE-4 inhibition results in increased intracellular cyclic adenosine monophosphate (cAMP) levels. The specific mechanism(s) by which crisaborole exerts its therapeutic action for the treatment of atopic dermatitis is not well defined. (PI §12.1, Ref ID 4027634)
| Trial Name | Design | N Randomised | Duration | Comparator | Primary Endpoint |
|---|
| AN2728-AD-301 (Trial 1) | Phase 3, R, DB, PC, parallel-group, multicenter (48 US sites) | 759 (Crisa 503 : Veh 256) | 28 days | Vehicle ointment BID | ISGA success at Day 29 (ITT, MI) |
| AN2728-AD-302 (Trial 2) | Phase 3, R, DB, PC, parallel-group, multicenter (identical design) | 763 (Crisa 513 : Veh 250) | 28 days | Vehicle ointment BID | ISGA success at Day 29 (ITT, MI) |
| AN2728-AD-303 | Phase 3, open-label, long-term safety, multicenter | 517 | 48 weeks (intermittent) | None (OL) | Safety/tolerability (no formal efficacy endpoint) |
| AN2728-AD-203 (MUSE PK) | Phase 2/PK, OL, maximal-use | 33 | 8 days | None | PK characterisation (Cmax, AUC₀₋₁₂) |
| Drug (INN) | Brand | Target / Class | Formulation | US Approval | Approved Population |
|---|
| Hydrocortisone / TCS (multiple) | Various | Glucocorticoid receptor | Cream/ointment (multiple strengths) | Various | Varies; some ≥3 months |
| Tacrolimus | Protopic | Calcineurin inhibitor (TCI) | Ointment 0.03%, 0.1% | December 2000 | ≥2 years (0.03%); moderate–severe AD; second-line |
| Pimecrolimus | Elidel | Calcineurin inhibitor (TCI) | Cream 1% | December 2001 | ≥2 years; mild–moderate AD; second-line |
| Crisaborole | EUCRISA | PDE-4 inhibitor | Ointment 2% | December 14, 2016 | ≥2 years; mild–moderate AD |
| Proprietary Name | EUCRISA™ |
| INN / Generic Name | crisaborole |
| Development Code | AN2728 |
| Manufacturer / Applicant | Anacor Pharmaceuticals, Inc., Palo Alto, CA 94303 USA |
| Pharmacologic Class | Phosphodiesterase 4 (PDE-4) inhibitor; boron-based benzoxaborole |
| Molecular Formula & MW | C₁₄H₁₀BNO₃; 251.1 g/mol |
| Chemical Name | 5-(4-cyanophenoxy)-1,3-dihydro-1-hydroxy-[2,1]-benzoxaborole |
| Dosage Form & Strength | Ointment, 2% (20 mg crisaborole per gram) |
| NDC — 60 g tube | 55724-211-21 |
| NDC — 100 g tube | 55724-211-11 |
| Inactive Ingredients | White petrolatum, propylene glycol, mono- and di-glycerides, paraffin, butylated hydroxytoluene, edetate calcium disodium |
| Storage | 20–25°C (68–77°F); excursions permitted 15–30°C (59–86°F) [USP Controlled Room Temperature]. Keep tube tightly closed. |
| Boxed Warning | None |
| PI Revision Date | December 2016 (Ref ID 4027634) |
Pooled Pivotal Trials (AN2728-AD-301 + AN2728-AD-302): Combined ITT N=1,522 across two identically designed trials. Randomised 2:1 (crisaborole:vehicle). 86.3% of subjects were 2–17 years of age. Demographic and baseline disease characteristics were comparable between treatment arms in both trials.
| Parameter | Crisaborole 2% BID (N=503) | Vehicle BID (N=256) |
|---|
| Age range (years) | 2–79 | 2–79 |
| Age 2–11 years, approx. % | ~60% | ~60% |
| Age 12–17 years, approx. % | ~25% | ~25% |
| Age ≥18 years, approx. % | ≤15% | ≤15% |
| Sex: Female | Majority | Majority |
| Race: White | Majority | Majority |
| Ethnicity: Not Hispanic/Latino | Majority | Majority |
| ISGA 2 (Mild) at baseline, % (pooled) | 38.5% | 38.5% |
| ISGA 3 (Moderate) at baseline, % (pooled) | 61.5% | 61.5% |
| Mean treatable BSA, % | ~18% | ~18% |
| Treatable BSA range | 5%–95% | 5%–95% |
| Prior biologic therapy | Excluded per protocol | Excluded per protocol |
| Parameter | Crisaborole 2% BID (N=513) | Vehicle BID (N=250) |
|---|
| Mean age (years) | 12.6 | 11.8 |
| Age range (years) | 2–79 | 2–79 |
| Age 2–11 years, approx. % | ~60% | ~60% |
| Age 12–17 years | ~25% | ~25% |
| Age ≥18 years | ≤15% | ≤15% |
| Sex: Female | Majority | Majority |
| Race: White | Majority | Majority |
| Ethnicity: Not Hispanic/Latino | Majority | Majority |
| ISGA 3 (Moderate) at baseline | Majority | Majority |
| Mean treatable BSA, % | 18% | 18% |
| Treatable BSA range | 5%–95% | 5%–95% |
| Dosing compliance (≥80–120% doses applied) | 93.6% | 87.6% |
| Category | Criterion |
|---|
| Inclusion — Age | Male or female ≥2 years at Baseline/Day 1 |
| Inclusion — Diagnosis | Clinical diagnosis of AD per Hanifin and Rajka criteria |
| Inclusion — Severity | ISGA score of Mild (2) or Moderate (3) at Baseline/Day 1 |
| Inclusion — BSA | AD involvement ≥5% treatable BSA (excluding scalp) |
| Inclusion — Sub-group quotas | ≥20% subjects aged 2–6 years; ≤15% subjects ≥18 years (per Division recommendation) |
| Exclusion — Disease stability | Unstable AD or consistent requirement for high-potency TCS to manage signs/symptoms |
| Exclusion — Infection | Significant active systemic or localised infection, including known actively infected AD |
| Exclusion — Prior treatment | Any history of biologic therapy including intravenous immunoglobulin |
| Exclusion — Concomitant treatment | Recent or anticipated concomitant use of systemic or topical therapies that might alter the course of AD |
Pre-specified subgroup analyses (primary endpoint): Gender, age group (2–11, 12–17, ≥18 years), race (White, Black, Other), ethnicity (Hispanic/Latino vs. not), and baseline ISGA (2 vs. 3). Per FDA Statistical Review (Matthew Guerra, PhD, dated 8/19/2016): treatment effect was greater in females than males, greater in White vs. Black subjects, and greater in subjects with moderate (ISGA 3) vs. mild (ISGA 2) disease at baseline. Treatment effect was greater in Hispanic/Latino vs. non-Hispanic subjects. Age subgroup results were inconsistent across the two trials. Source: FDA Medical Review NDA 207695 (Ref ID 3983007), Tables 26–27, p. 90.
Primary Endpoint: Proportion of subjects achieving ISGA success at Day 29 — defined as ISGA score of Clear (0) or Almost Clear (1) with ≥2-grade improvement from baseline. Statistical framework: Logistic regression with Firth’s penalised likelihood; factors = treatment group + analysis centre. Missing efficacy data: Markov Chain Monte Carlo (MCMC) multiple imputation (MI). Hypothesis testing: two-sided α=0.05. FWER controlled sequentially (primary → secondary ISGA 0/1 → time to ISGA success).
Crisaborole 2% BID (N=503)
p=0.038
Crisaborole 2% BID (N=513)
p<0.001 Data availability note: Intermediate timepoint values (Days 1, 8, 15, 22) are presented graphically only in FDA PI Figure 1 and Medical Review Figure 5; they are not reported in extractable tables. Per data accuracy standards, only the explicitly tabulated Day 29 values are plotted. Intermediate points marked null (spanGaps: false).
ISGA Success Rate (%) at Day 29 — Trials 1 & 2 (ITT, MI)
Source: FDA PI NDA 207695 (Ref ID 4027634), Table 2 (§14); FDA Medical Review NDA 207695 (Ref ID 3983007), Table 17. Only Day 29 values are explicitly tabulated; intermediate timepoints not plotted per data accuracy standards.
Trial 1 — Crisaborole (N=503)
p=0.005
Trial 1 — Vehicle (N=256)
Trial 2 — Crisaborole (N=513)
p<0.001
Trial 2 — Vehicle (N=250) | Endpoint | Trial 1 — Crisa / Vehicle | p-value | Trial 2 — Crisa / Vehicle | p-value |
|---|
| ISGA success Day 29 (primary, ITT/MI) | 32.8% / 25.4% | 0.038 | 31.4% / 18.0% | <0.001 |
| ISGA Clear or Almost Clear Day 29 (2°, ITT/MI) | 51.7% / 40.6% | 0.005 | 48.5% / 29.7% | <0.001 |
| Time to ISGA success (2°, log-rank) | Median NC | <0.001 | Median NC | <0.001 |
| ISGA success Day 29 (PP population) | 32.4% / 26.9% | 0.088a | 32.2% / 18.3% | <0.001 |
| ISGA Clear or Almost Clear (PP) | 51.7% / 43.8% | 0.032 | 50.0% / 29.8% | <0.001 |
| Time to ISGA success (PP, log-rank) | Median NC | 0.003 | Median NC | <0.001 |
| PRO/Endpoint | Instrument / Scale | Finding | Regulatory Status |
|---|
| AD signs improvement (erythema, induration/papulation, exudation, excoriation, lichenification) | 4-point global assessment scale; success = None (0) or Mild (1) with ≥1-grade improvement from baseline | Greater proportion of crisaborole subjects improved on all signs vs. vehicle at Day 29 (both trials) | Exploratory — FDA-agreed not for labelling (PRO dossier adequate for exploratory; EOP2 guidance) |
| Time to pruritus improvement | 4-point pruritus severity scale; success = None (0) or Mild (1) with ≥1-grade improvement | Greater improvement in crisaborole arm at Day 29 (both trials) | Exploratory — downgraded from secondary; baseline pruritus PRO dossier not submitted pre-NDA |
No Boxed Warning. EUCRISA (crisaborole) ointment, 2% carries no boxed warning. This is a key differentiator from topical calcineurin inhibitors (tacrolimus, pimecrolimus) which carry a boxed warning regarding potential malignancy risk. (PI §1, Ref ID 4027634)
Safety Population (Controlled)
1,511
Crisa N=1,012; Vehicle N=499 (pooled Trials 301+302)
Long-Term Safety N
517
AN2728-AD-303; up to 48 weeks intermittent
Follow-up Duration
28 days
Pivotal trials; 48 weeks in LT trial (52.4% completed 12 mo)
Most Common ADR (≥1%)
4.0%
Application site pain (burning/stinging) — crisa vs. 1.0% vehicle
- Hypersensitivity Reactions (§5.1): Hypersensitivity reactions including contact urticaria have occurred. Hypersensitivity should be suspected in the event of severe pruritus, swelling, and erythema at the application site or at a distant site. If signs and symptoms occur, discontinue EUCRISA immediately and initiate appropriate therapy.
| Adverse Reaction | Crisaborole 2% BID (N=1,012) n (%) | Vehicle BID (N=499) n (%) |
|---|
| Application site paina | 45 (4%) | 6 (1%) |
| Trial | Crisaborole n (%) | Vehicle n (%) | Notable SAE Terms |
|---|
| AN2728-AD-301 | 5/502 (1.0%) | 1/252 (0.4%) | Appendicitis, Kawasaki’s disease, suicide attempt, pneumonia, acute asthma exacerbation — all assessed unrelated |
| AN2728-AD-302 | 3/510 (0.6%) | 0/247 (0%) | Multiple lacerations (accident), impetigo at application site, suicidal ideation — none clearly related |
| AN2728-AD-303 (LT, OL) | Multiple (see narrative) | N/A (OL) | Upper respiratory infection, eczema superinfection, asthma exacerbation, MRSA — none drug-related |
No deaths were reported in either pivotal Phase 3 controlled trial (AN2728-AD-301, AN2728-AD-302) or in the long-term safety trial (AN2728-AD-303). Source: FDA Medical Review NDA 207695 (Ref ID 3983007).
| Trial / Population | Crisaborole | Vehicle | Leading AE Terms (Crisa arm) |
|---|
| Pooled Trials 301+302 (Safety Pop.) | 12/1,021 (1.2%) | 6/506 (1.2%) | Majority: application site reactions. Age 2–11 predominant (10/12 subjects). No subject ≥18 years discontinued due to AE. |
| AN2728-AD-303 (LT Safety) | 9/517 (1.7%) | N/A | Dermatitis atopic (5, 1.0%), application site pain (2, 0.4%), application site dermatitis (1, 0.2%), eczema (1, 0.2%) |
| Adverse Event (MedDRA PT) | Crisaborole 2% BID (N=1,012) n (%) | Vehicle BID (N=499) n (%) |
|---|
| Application site pain | 8 (0.8%) | 0 (0%) |
| Dermatitis atopic | 3 (0.3%) | 2 (0.4%) |
| Pruritus | 2 (0.2%) | 1 (0.2%) |
No clinically meaningful laboratory abnormalities were identified in the pivotal controlled trials. Standard serum chemistry and haematology were assessed at Screening, Baseline, and Day 29. No drug-related laboratory signals were reported in the Medical Review. Source: FDA Medical Review NDA 207695 (Ref ID 3983007), Section 8.
Thorough QT Study (AN2728-TQT-108): At doses up to 45 g/day (~60% BSA), no clinically relevant QTc prolongation observed. No subject had QTcF >480 ms; no change in QTcF >60 ms. In Phase 3 ECG subset (N=615), 2 crisaborole subjects aged ≥18 had QTcF 450–480 ms.
QT-IRT caveat: TQT exposures did not cover exposures seen in the Phase 3 pediatric PK study (mean Cmax 127 ng/mL at Day 8; individual Cmax up to 1,170 ng/mL in age 6–11 subgroup). QT effect in the very young, high-BSA population cannot be fully excluded from available data. Source: FDA Medical Review NDA 207695 (Ref ID 3983007), Sections 4.5.2 and 8.4.9.
Long-term safety trial AN2728-AD-303 (N=517): 12 subjects (2.3%) reported events in psychiatric disorders SOC: anxiety (2, 0.39%), ADHD (4, 0.77%), depression (4, 0.77%), insomnia (2, 0.39%), suicidal ideation (1, 0.19%), suicide attempt (1, 0.19%). Only 1 subject was ≥18 years. No correlation between greater drug exposure and greater psychiatric events. Pivotal controlled trials: 1 suicide attempt (Trial 301, crisaborole arm, Day 27 — assessed unrelated; onset of stress 2 months prior); 1 suicidal ideation (Trial 302, vehicle arm). Source: FDA Medical Review NDA 207695 (Ref ID 3983007), Table 55, Section 8.11.
| Population | Data Available | Key Findings |
|---|
| Pregnancy | No human data | Animal: no adverse fetal effects at ≤5× MRHD (rat) and ≤3× MRHD (rabbit). Maternal toxicity at 18× MRHD associated with stillbirths, pup mortality, reduced pup weights. No fetal malformations noted. |
| Lactation | No data (human milk) | Systemically absorbed. No data on breast milk presence or infant effects. Weigh breastfeeding benefits vs. potential risk. |
| Paediatric ≥2 years | 1,313 paediatric subjects (pivotal RCTs) | Safety and efficacy established. 86.3% of pivotal trial subjects were aged 2–17 years. |
| Paediatric <2 years | Not established | Post-marketing deferred study required (open-label safety/PK; ages 3 months–<2 years; N≥100 evaluable). |
| Geriatric (≥65 years) | Insufficient numbers | Clinical studies did not include sufficient subjects ≥65 years to determine age-specific differences in response. |
| Renal/Hepatic Impairment | No formal studies | No dosing recommendations in label. Topical route with low systemic exposure. |
Carcinogenicity (Oral, Sprague-Dawley Rat)
Drug-related increased incidence of benign granular cell tumors in uterus with cervix or vagina (combined) at 300 mg/kg/day (2× MRHD on AUC basis). Clinical relevance unknown. No neoplastic findings in dermal study (CD-1 mice) up to 7% crisaborole (2× MRHD).
Genotoxicity
Negative in all assays: Ames assay (in vitro); human lymphocyte chromosomal aberration assay (in vitro); rat micronucleus assay (in vivo). No mutagenic or clastogenic potential identified.
Reproductive Toxicity / Fertility
No effects on fertility in male or female rats at up to 600 mg/kg/day (18× MRHD). Maternal toxicity at 600 mg/kg/day in prenatal/postnatal rat study: stillbirths, pup mortality, reduced pup weights. No fetal malformations.
Cardiac Safety (hERG)
Crisaborole classified as low-potency hERG channel blocker in vitro. Safety pharmacology studies in rats, mice, dogs, minipigs: no cardiac effects at therapeutic exposures. One dog death at 300 mg/kg/day (high-dose); no QTc effects at that dose level. TQT study negative at 45 g/day.
| Treatment-Related Adverse Reaction | Crisaborole 2% BID (N=517) | Causality |
|---|
| Any treatment-related AE | 34 (6.6%) | Possible/Probable/Definite combined |
| Dermatitis atopic | 11 (2.1%) possible; 4 (0.8%) probable; 1 (0.2%) definite | Possible/Probable/Definite |
| Application site pain | 3 (0.6%) possible; 7 (1.4%) probable; 6 (1.2%) definite | Possible/Probable/Definite |
| Application site infection | Not reported at ≥1% | — |
Weight loss signal (AN2728-AD-303): Clinically meaningful weight loss (>10% at any timepoint) was observed in a subset of subjects, particularly in the 2–9 year age group. A correlation between weight change from baseline and total crisaborole usage (g) was observed in the 2–9 year cohort (Figure 7, Medical Review). No similar signal in the 28-day controlled trials. Clinical relevance uncertain. Monitored in post-approval safety programme. Source: FDA Medical Review NDA 207695 (Ref ID 3983007), Tables 58–61, Figures 6–7.
Cmax (Day 8)
127 ± 196
ng/mL mean ± SD; pediatric AD subjects (n=33); at steady state
AUC₀₋₁₂ (Day 8)
949 ± 1240
ng·h/mL mean ± SD; 0–12 h post-dose; steady state
Steady State
Day 8
Systemic concentrations at steady state by Day 8 (BID × 8 days)
Accumulation (Parent)
1.9×
AUC₀₋₁₂ ratio Day 8/Day 1
Plasma Protein Binding
97%
In vitro; human plasma proteins
Route of Elimination
Renal
Renal excretion of inactive metabolites is the major route of elimination
Percutaneous absorption: Plasma concentrations were quantifiable in all 33 pediatric subjects (BSA involvement 27–92%) following topical application of approximately 3 mg/cm² BID. Dose range per application was approximately 6–30 g. Systemic concentrations reached steady state by Day 8, with a mean accumulation factor of 1.9 for the parent compound. Quantifiable systemic exposure confirms that crisaborole is systemically absorbed through affected skin. No food-effect study applicable (topical route). (PI §12.3, Ref ID 4027634)
Crisaborole is 97% bound to human plasma proteins based on in vitro studies. Volume of distribution data not explicitly reported in the PI or Medical Review for the topical formulation. (PI §12.3, Ref ID 4027634)
| Metabolite | Pathway | Accumulation Factor (Day 8/Day 1) | Activity |
|---|
| Metabolite 1: 5-(4-cyanophenoxy)-2-hydroxyl benzylalcohol (AN7602) | Hydrolysis of parent crisaborole | 1.7× | Inactive |
| Metabolite 2: 5-(4-cyanophenoxy)-2-hydroxyl benzoic acid | Oxidation of Metabolite 1 | 6.3× (significant accumulation) | Inactive |
Crisaborole is substantially metabolised into inactive metabolites. Both Metabolite 1 and Metabolite 2 were at or near steady state by Day 8. The high accumulation factor for Metabolite 2 (6.3×) was flagged in FDA review as requiring in vitro CYP DDI evaluation. (PI §12.3, FDA Medical Review §5, Ref ID 3983007)
| CYP Enzyme | Crisaborole / Met-1 (in vitro) | Metabolite 2 (in vitro) | Clinical DDI Study | Recommendation |
|---|
| CYP1A2, 2B6 | No inhibition | Weak inhibitor | None conducted | Not clinically relevant at therapeutic exposures |
| CYP2C8, 2C9 | No inhibition | Moderate inhibitor (in vitro) | Warfarin (CYP2C9 substrate) — no DDI observed | No dose adjustment required |
| CYP2C19, 2D6, 3A4 | No inhibition | No inhibition | None conducted | No concern |
| CYP induction (all) | No induction (parent + Met-1 + Met-2) | No induction | None conducted | No concern |
| Population | Data | Key Finding |
|---|
| Paediatric 2–17 years | PK study AN2728-AD-203 (n=33) | Cmax 127 ± 196 ng/mL; wide inter-subject variability (CV ~154%). Age 6–11 subgroup: highest mean Cmax ~205 ng/mL; individual Cmax up to 1,170 ng/mL. Steady state Day 8. |
| Adults / Elderly | Not formally studied (topical) | Not reported. ≤15% of pivotal trial subjects were adults; no PK sub-study in adults. |
| Renal impairment | Not formally studied | Not reported. Low systemic exposure; no dose adjustment recommended. |
| Hepatic impairment | Not formally studied | Not reported. No dose adjustment recommended. |
| BSA effect | PK study (range 27–92% BSA) | Wide range of BSA involvement studied; systemic exposure increased with increasing BSA involvement (high variability). |
PDE-4 inhibition via boron-based scaffold: Crisaborole is a novel benzoxaborole compound in which the boron atom within a cyclic boronate ester provides selective, reversible binding to PDE-4 through interaction with the bidentate chelation site of the catalytic domain. PDE-4 is the primary cyclic AMP-hydrolysing enzyme in inflammatory cells including T lymphocytes, eosinophils, mast cells, and keratinocytes. Inhibition of PDE-4 elevates intracellular cAMP, leading to protein kinase A activation and downstream suppression of pro-inflammatory cytokine production including IL-4, IL-13, IL-31, TNF-α, and IFN-γ. The precise mechanism(s) by which these effects translate to clinical improvement of atopic dermatitis has not been fully elucidated. (PI §12.1, Ref ID 4027634; FDA Medical Review §4, Ref ID 3983007)
QTc pharmacodynamics: At therapeutic doses, EUCRISA ointment is not expected to prolong QTc to any clinically relevant extent based on thorough QT study AN2728-TQT-108 at doses up to 45 g/day (~60% BSA). No formal PK/PD modelling for ISGA response vs. plasma exposure was reported in the available FDA documents. Dose–response relationship was not formally assessed in the Phase 3 design (single dose level). (PI §12.2, Ref ID 4027634)
Approved Dose
2%
Thin layer applied BID to all affected areas
Route
Topical
External use only; not ophthalmic, oral, or intravaginal
Frequency
BID
Twice daily; no specific time interval specified
Duration
As needed
No defined maximum treatment duration in label
Standard Dosing
- Apply a thin layer of EUCRISA to all affected areas twice daily
- No loading dose; no titration schedule
- Single approved dose: 2% ointment (no lower strength available)
- Approved for patients ≥2 years; same dosing regimen across all age groups ≥2 years
- No weight-based dose calculation required
Dose Modifications
- Renal impairment: No dose adjustment recommended (no formal PK study; topical route)
- Hepatic impairment: No dose adjustment recommended (no formal PK study)
- Age ≥65 years: Insufficient data — no dose adjustment specified in label
- Body weight: No weight-based adjustment
- Paediatric <2 years: Not established — do not use
Preparation & Administration
- No reconstitution required (ready-to-use ointment)
- Apply to affected skin areas; avoid eyes, mouth, and vagina
- No restriction on application site (apply to all affected areas including face, neck, folds, extremities)
- Wash hands after application unless hands are the treated area
- No restrictions on concomitant use of emollients stated in label; apply emollients separately
- Caregiver: wash hands after applying to another person
Missed Dose / Storage
- Missed dose: No specific instructions in PI; apply as soon as remembered and resume regular schedule
- Store at 20–25°C (68–77°F)
- Excursions permitted: 15–30°C (59–86°F) [USP Controlled Room Temperature]
- Keep tube tightly closed
- Available as 60 g and 100 g laminate tubes
Contraindication (PI §4): EUCRISA is contraindicated in patients with known hypersensitivity to crisaborole or any component of the formulation. Source: FDA PI NDA 207695 (Ref ID 4027634), §4.
Hypersensitivity monitoring: Monitor for signs of hypersensitivity reactions at or distant from the application site (severe pruritus, swelling, erythema). No pre-treatment laboratory monitoring or screening required. No vaccination precautions applicable (non-immunosuppressive mechanism). No TB screening requirement.
No dose adjustments required for drug interactions. CYP2C9 clinical DDI study with warfarin showed no interaction. Metabolite 2 is a moderate in vitro CYP2C8/2C9 inhibitor but no clinically meaningful interaction was demonstrated at therapeutic exposures. No DDI-based dose modifications recommended in PI. Source: FDA PI NDA 207695 (Ref ID 4027634), §12.3.
| Parameter | Recommendation |
|---|
| Application technique | Apply a thin layer with gentle spreading over affected areas; no rubbing instruction specified |
| Application areas | All affected skin areas (no anatomical restrictions stated in label). Not for ophthalmic, oral, or intravaginal use. |
| Concomitant emollients | No specific instructions in PI; no restrictions stated |
| Hand hygiene | Wash hands after applying unless hands are the treatment area. Caregivers must also wash hands after application. |
| Occlusion | Not addressed in PI |
| Paediatric administration | Caregiver-applied acceptable; same technique as adult self-application |
| Application Number | NDA 207695 |
| Application Type | NDA 505(b)(1) — original NDA; New Molecular Entity (NME) |
| Applicant | Anacor Pharmaceuticals, Inc., Palo Alto, CA 94303 |
| Original Developer | Anacor Pharmaceuticals (acquired by Pfizer Inc. in 2016 during NDA review) |
| Submission Date | January 7, 2016 |
| PDUFA Goal Date | January 6, 2017 |
| Actual Approval Date | December 14, 2016 |
| Review Division | Division of Dermatology and Dental Products (DDDP), CDER |
| Review Type | Standard Review |
| Breakthrough Therapy Designation | Not granted |
| Fast Track Designation | Not reported in available documents |
| Orphan Drug Designation | Not applicable (atopic dermatitis is not an orphan indication) |
| Advisory Committee | Not convened (no AdCom for this NDA) |
| Medical Reviewer | Melinda L. McCord, MD (Review completion date: September 7, 2016) |
| Medical Review Ref ID | 3983007 |
| PI Ref ID | 4027634 |
2012–2013 (ESTIMATED)
IND Programme Established
Phase 1/2 programme initiated with crisaborole (AN2728) in psoriasis and atopic dermatitis. Phase 2 trials used intra-subject lesion-level comparisons with ADSI endpoints — acknowledged as limited for informing Phase 3 assumptions.
FEBRUARY 26, 2014
End-of-Phase 2 (EOP2) Meeting
Division agreed to ISGA-based primary endpoint (ISGA 0/1 with ≥2-grade improvement). Recommended: adult enrolment ≤15%; age 2–6 subgroup ≥20%; EASI replaced by global AD sign assessment; pruritus PRO downgraded to exploratory pending dossier. Applicant accepted all recommendations.
JUNE 2014
Advice Letter (Cardiac Safety)
Division recommended addition of ECG assessment at Baseline and Day 8 at selected Phase 3 sites; full physical examination at end of treatment; enhanced safety monitoring per advice letter June 16, 2014.
2014–2015
Phase 3 Pivotal Trials Conducted (AN2728-AD-301 and AN2728-AD-302)
Two identically designed multicenter trials (48 US sites each). Long-term safety trial AN2728-AD-303 initiated concurrently.
SEPTEMBER 23, 2015
Pre-NDA Meeting
FDA requested in vitro CYP inhibition/induction studies for major metabolite AN7602 (Metabolite 1/2) — results from PK study showed exposure >30% of parent. Sponsor agreed to add warfarin DDI study to NDA package.
JANUARY 7, 2016
NDA 207695 Submitted
Standard NDA submission. Data package included AN2728-AD-301, AN2728-AD-302, AN2728-AD-303 (interim), and multiple PK/safety pharmacology studies including warfarin DDI.
APRIL–MAY 2016
FDA Site Inspections
Four sites inspected (Centers 138, 150, 211, 240). Centers 150, 211, 240: NAI (No Action Indicated). Center 138: VAI (Voluntary Action Indicated). Sensitivity analysis excluding Center 138 showed only minor change in response rates — no impact on data integrity.
SEPTEMBER 7, 2016
Medical Review Completed — Recommendation for Approval
Medical reviewer Melinda McCord, MD recommended approval pending labelling negotiations. Benefit-risk: modest but statistically significant efficacy; favourable safety profile; first non-steroidal/non-TCI topical option for mild-moderate AD without malignancy boxed warning.
DECEMBER 14, 2016
FDA Approval — NDA 207695
EUCRISA (crisaborole) ointment, 2% approved for topical treatment of mild to moderate atopic dermatitis in patients 2 years of age and older. First PDE-4 inhibitor approved for atopic dermatitis in the United States. Approved ahead of PDUFA date (January 6, 2017).
Effect Size and Statistical Significance (Trial 301 PP)
Trial 301 primary endpoint failed to reach statistical significance in the Per Protocol population (p=0.088). FDA accepted this based on: (1) confirmed significance in Trial 302 ITT and PP; (2) identical trial designs (pre-specified replicate strategy); (3) significance on both PP secondary endpoints in Trial 301. Modest absolute effect size (~7–13 percentage points over vehicle) was acknowledged but accepted given non-steroidal mechanism and chronic disease context.
Phase 2 Estimates for Phase 3 Power Calculations
FDA noted Phase 3 power was not based on Phase 2 data using the same study population, endpoints, or success definition. Phase 2 used intra-subject comparisons and ADSI (different from ISGA success). Division accepted the design but flagged this limitation in the EOP2 review.
Metabolite 2 CYP Inhibition — In vitro Signal
Metabolite 2 showed moderate in vitro inhibition of CYP2C8 and CYP2C9. As the most sensitive enzyme (CYP2C9), a clinical DDI study using warfarin as a probe substrate was required. Results demonstrated no clinically meaningful drug interaction at therapeutic exposures. Resolved in NDA.
Cardiac Safety / QTc Exposure Gap
TQT study (AN2728-TQT-108) performed at up to 45 g/day did not achieve exposures comparable to those observed in the Phase 3 pediatric PK study (individual Cmax up to 1,170 ng/mL in age 6–11 subjects). QT-IRT acknowledged no signal but flagged residual uncertainty in high-BSA pediatric subjects. No additional requirements mandated at approval.
Psychiatric AE Signal — Suicidality
Two suicidal ideation/attempt events in the pivotal trials (one in each arm). Neither was attributed to crisaborole. AD itself is a risk factor for psychiatric comorbidity. Long-term trial AN2728-AD-303 showed low rates of psychiatric events (2.3%) with no exposure correlation. No labelling requirement for suicidality monitoring.
Paediatric <2 Years — Deferred Study
Safety and efficacy in children <2 years not established. Post-marketing study deferred because EUCRISA was ready for approval in ≥2 years population. PMR required: open-label safety/PK study in ages 3 months–<2 years (N≥100 evaluable; N≥16 moderate AD with BSA ≥35% for PK sub-study).
Post-Marketing Requirement (PMR): One post-marketing study required as a condition of approval per Paediatric Research Equity Act (PREA) deferral.
| Study | Type | Objective | Key Design Elements |
|---|
| Paediatric safety and PK study (ages 3 months–<2 years) | PMR (PREA deferral) | Establish safety and PK in infants/toddlers <2 years | Open-label; N≥100 evaluable (safety); N≥16 evaluable moderate AD + BSA ≥35% (PK sub-study, maximal use); mild-to-moderate AD; treatable BSA ≥5% |
| Domain | Note |
|---|
| Benefit-risk conclusion | Favourable. First non-steroidal, non-calcineurin inhibitor topical option for mild-moderate AD without malignancy boxed warning. Modest efficacy vs. vehicle accepted in context of chronic paediatric disease, clean safety profile, and unmet need for alternative mechanisms. |
| Labelling negotiations | FDA removed applicant-proposed language (redacted §(b)(4)) from §14 Clinical Studies; revised success rate description; added Figure 1 time-course chart. INN used throughout body text; brand name only in header. |
| Immunogenicity | Not applicable (small molecule topical agent). |
| Statistical issues | MCMC multiple imputation (140 datasets for Trial 301; 135 for Trial 302). Site 138 financial disclosure (investigator Anacor stock >$50,000): sensitivity analysis showed negligible impact on results. See FDA Statistical Review, Matthew Guerra, PhD, dated 8/19/2016. |
| Pharmacovigilance | Standard FDA pharmacovigilance; no REMS required. Post-marketing monitoring for weight change signal in young children (2–9 years) and psychiatric AEs. |
| Source Documents | FDA PI: Ref ID 4027634 (Revised 12/2016). FDA Medical Review: Ref ID 3983007 (NDA 207695, Melinda McCord MD, 09/07/2016). |